What’s next for high-tech nappy maker Simavita

Pic: Charnchai / iStock / Getty Images Plus via Getty Images
Like most biotech stocks, Simavita boasts a huge market: the $US64 billion a year ($A88 billion) global infant and adult nappy sector.
But like all-too-many biotechs, the incontinence management play has been frustratingly slow in generating revenue, despite regularly returning to investors for top-up funds including a $3 million whip-around announced on October 3.
The original Simavita (ASX:SVA) business consists of wi-fi-enabled products that detect urinary incontinence and then monitor and chart patient progress.
The premise is that nurses and attendants at aged care facilities spend at least 25 per cent of their time dealing with incontinence issues, with 70 per cent of residents affected.
The condition costs such facilities $1.1 billion a year, with a further $112 million expended on incontinence products annually, the Australian Health and Welfare Institute estimates.
But after a board and management change in 2016 and a strategy change (dubbed Pivot) in early 2017, the company is pinning its hopes on the broader nappy market.
Simavita is predicated on the razor blade model — it makes money out of the consumables (the sensors embedded in the nappy during manufacturing) and not so much out of the one-off hardware and software.
While the world consumes 212 billion infant nappies and 27 billion adult nappies annually, the branded manufacturers are being challenged by the private label market and need a point of differentiation.
Not surprisingly, the market is also price sensitive.
“As long as it doesn’t leak, people will continue to buy the same product,” chief commercial officer Peta Jurd says.
Having sold third-party products previously, Amazon recently launched its own price-sensitive brand and is the third biggest seller in the US.
Simavita’s painful past
Simavita’s technology was devised by an Australian doctor, the late Dr Fred Bergman, while working at a Melbourne residential aged care facility. He filed for a patent in 1996 and by 2008 had developed four iterations of the device.
Along the way, he attracted $28 million of funding from private money and government grants.
Simavita back-door listed via the shell of Genetic Technologies subsidiary Gtech International Resources in 2014, raising $1.1 million by issuing 2.7 million shares — or technically “CHESS Depositary Interests” (CDIs) which are a proxy for trading foreign shares on the ASX.
Why CDIs? Gtech was actually listed on the Toronto Stock Exchange.
By 2016 it was clear things were far from dandy and the board instituted a program to halve costs.
This included then chief executive officer Philippa Lewis assenting to a 30 per cent pay cut (her salary package of $1.6 million in 2013-14 — including $734,000 of share-based options — did not exactly go unnoticed by investors).
By April 2016 Ms Lewis and chairman Michael Brown had agreed to step down, replaced by Mesoblast director Michael Spooner and Resmed and Antisense Therapeutics director Gary Pace.
Around this time three other directors departed: Damian Haakman, Ari Bergman (Fred’s son) and Craig Holland.
As part of the cost purge the company delisted from the Toronto bourse in July 2016.
“The company has had an interesting history — that’s one way of describing it,” Ms Jurd says.
“There’s been a lot of money invested in it but we have a very substantial robust patent portfolio.”
Simavita’s products
Simavita’s first product, SIM (smart incontinence monitor) Clinical was approved in the US in 2013 as a class II medical device. It was also approved in Europe as a class I device.
Based on an inserted sensor, SIM developed an incontinence profile of patients to help with toileting regimens.
In late 2016 the company launched a portable variant for home care and institutional use called Assessplus. The product is approved for funding under the National Disability Insurance Scheme.
Simavita is now developing Alertplus, which monitors wetness via sensors printed into the nappy. The sensors are read by a low-cost, multi-use device that clips on to the nappy and connects to a phone app via Bluetooth.
The company estimated each sensor will cost a mere one cent to make, with the receiver devices also costing one cent per use over a six-month life.
Along the way, the company abandoned its direct sales model, in favour of partnerships.
Under the new management, the company stopped trying to sell monitor nappies to aged care homes, changing the business model to embedding monitors in commercial nappy production.
- Subscribe to our daily newsletter
- Bookmark this link forsmall cap news
- Join our small cap Facebook group
- Follow us on Facebook or Twitter
The premise is that the “dumb diaper” market hasn’t changed in a quarter of a century and is highly competitive. So, the first maker to introduce smart sensor technology at an affordable price will obtain a key advantage.
In August, Simavita announced its first big marketing tie up, with the Belgian nappy maker Drylock Technologies which specialises in the private label sector.
For a 12 months period, the parties will “use their best endeavours to seek out and identify tender opportunities with large mass market customers and to work exclusively with each other only in relation to agreed tender opportunities.”
More specifically, the parties are targeting four mass market customers, accounting for around EUR50 million ($80 million) each.
The company cites six potential licencing partners who make “billions” of nappies annually.
Value leakage
In its full-year accounts, directors opined about the “material uncertainty” of Simavita continuing as a going concern.
Happily, these concerns have been ameliorated by this month’s capital raising: a private placement of $1.975 million at 3.0 cents a share (a 20 percent discount to the 15-day average share price) and a $1 million convertible note.
The company also issued $1.3 million of convertible notes in April.
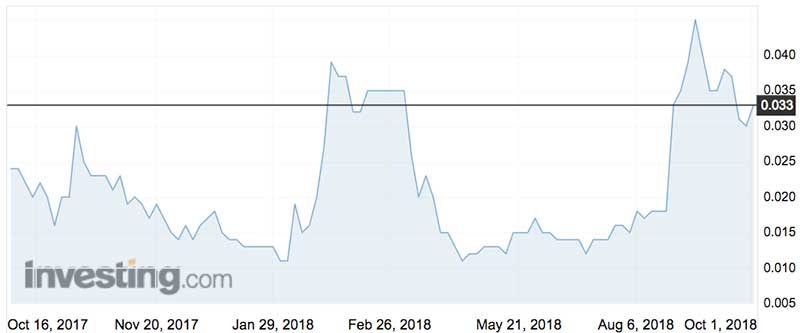
While Simavita is striving to keep nappies intact it’s been more a case of value leakage, with its shares losing more than 90 per cent of their value since listing five years ago.
Simavita shares have traded as high as 4.6c over the last 12 months (in late April) and as low as 1.1c (late August).
The company has ploughed $70 million into product development over its corporate life, $12 million of this in the past year.
Dr Boreham’s diagnosis
Simavita might fail the smell test – literally.
From your columnist’s fading recollection of rearing infants, it was kind of obvious when a nappy change was due — although maybe that was more in relation to “number twos”.
Dr Boreham is happy to be proved wrong, if and when Simavita snares a licensing deal with a major nappy maker.
In the aged care context, on paper it makes sense for larger facilities to invest in Assessplus, but given the low take-up it looks like inertia prevails.
Ms Jurd says Alert is a platform technology that in future could add temperature and hydration measurement and GPS tracking of elderly patients.
But without mandatory monitoring requirements for the aged care sector, Simavita is likely to continue to struggle for traction in this market.
Simavita’s material, by the way, promises “no more worries … no more messy diapers” which biologically is a big call indeed.
But if nappy makers can get away with thinner nappies that use less pulp, that’s a bottom-line win for the manufacturer, the parents and the environment.
Disclosure: Dr Boreham is not a qualified medical practitioner and does not possess a doctorate of any sort. But he thinks he has got to the bottom of this one anyway. The content of this article was not selected, modified or otherwise controlled by Stockhead. Stockhead has not provided, endorsed or otherwise assumed responsibility for any financial product advice contained in this article.
This article first appeared at biotechdaily.com.au
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








