Tax cut takes a bite out of Penthrox maker Medical Development’s profits

Tax cuts are great, right? Not always, as Medical Developments — maker of the famous Aussie “green whistle” pain reliever — pointed out in its 2018 results today.
MedDev’s Penthrox product — often referred to as the “green whistle” — is a self-administered non-opioid pain relief inhaler used in trauma settings.
It’s used in countries around the world — though a US roll-out plan just ground to a halt with authorities there suspending an application aimed at getting access to North America.
Today MedDev reported its full-year profit had plunged 87 per cent to $243,000 — and the reason they gave was a tax cut, from 30 per cent to 27.5 per cent.
The tax change meant they had to lower the value of a deferred tax asset and add a $107,000 charge to their expenses.
MedDev (ASX:MVP) also reported a 17 per cent rise in expenses and a drop in revenue of 5 per cent to $17.9 million.
The company blamed higher expenses on rising ‘pharmacovigilance’ costs as they push their non-opioid painkiller Penthrox into new countries and higher marketing and distribution expenses.
‘Pharmacovigilance’ is the extra monitoring of drugs after they have been licensed particularly to identify and previously unreported adverse reactions.
MVP said delays in getting Penthrox approved in 23 European countries until the end of 2018 meant sales have been pushed into 2019.
- Subscribe to our daily newsletter
- Bookmark this link forsmall cap news
- Join our small cap Facebook group
- Follow us on Facebook or Twitter
MVP is still waiting on the US FDA to release Penthrox’s New Investigational Drug application.
Sales in France, the UK, Ireland, New Zealand and Australia remained steady or rose, but fell in Singapore and South Africa.
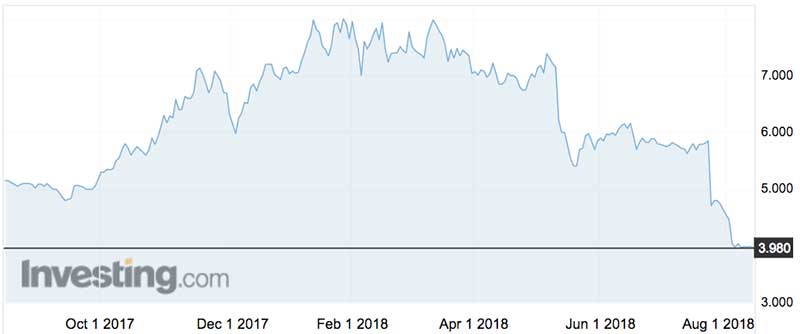
The company’s shares are expected to open slightly higher on Friday.
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








