Neuren shareholders throw their toys after licensing deal; shares plummet 45pc

Investors have decimated Neuren Pharma’s share price after the company signed a licensing deal that did not live up to expectations.
Neuren (ASX:NEU) — which develops therapies for brain injuries — signed a deal with NASDAQ-listed ACADIA Pharma to develop and commercialise its Rett syndrome treatment, a debilitating neurological disorder that occurs primarily in baby girls.
Investors had been hoping for a blockbuster deal.
But the $US10 million ($13.5 million) up-front payment did not live up to expectations — and sent shareholders off the deep end.
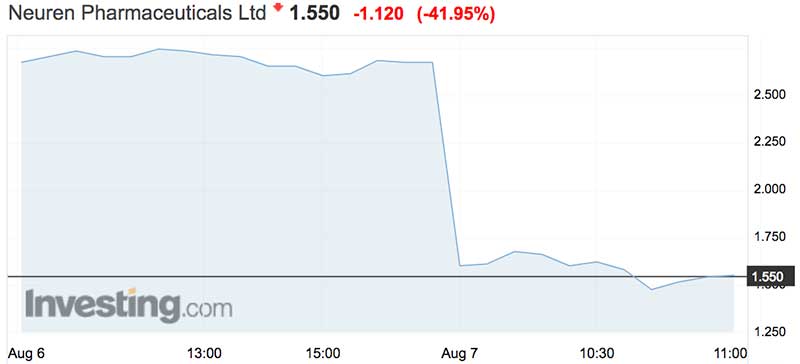
The stock plummeted 45 per cent to an intraday low of $1.47, it’s lowest point since October last year.
It recovered slightly to close at $1.52, valuing the company at about $265 million. The shares have traded between $1.16 and $3.60 over the past year.
Neuren flagged in May that it was in early licensing talks, sending its shares up 10 per cent.
At the time it said the then-unnamed US company would invest $US4 million ($5.3 million) for 1.3 million Neuren shares to pay for an exclusivity period.
It valued Neuren at $4 a share.
ACADIA will launch a Phase 3 study in the second half of 2019, evaluating Neuren’s drug trofinetide in about 180 girls with Rett syndrome.
The trial will cost $US55 million, paid for by ACADIA.
That $US4 million it paid in May will fund the preparations for the trial.
Potential milestone payments are worth up to $US455 million with royalties available on top of that.
Neuren’s market cap is $265 million.
It will retain rights to develop and commercialise trofinetide for all diseases outside of North America, such as Fragile X syndrome, a genetic defect that is a known cause of autism.
- Subscribe to our daily newsletter
- Bookmark this link for small cap news
- Join our small cap Facebook group
- Follow us on Facebook or Twitter
Trofinetide is designed to treat the core symptoms of Rett syndrome by reducing neuroinflammation and supporting synaptic function. It’s been granted FDA Fast Track Status and Orphan Drug Designation in the US and Europe for both Rett syndrome and Fragile X syndrome.
Stockhead is seeking comment from Neuren.
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








