Med-tech Admedus on track for profit after near-death experience, says CEO

Pic: Luis Alvarez / DigitalVision via Getty Images
Admedus is on track to break even by the end of the year — and make a profit in early 2019 — after coming close to insolvency last year, chief Wayne Paterson says.
The cardiac-focused med-tech is also close to finding a partner for its troublesome immunotherapy business.
Mr Paterson says he will “know either way” by the end of March if the company (ASX:AHZ) has a partner that will take the lead financially for the drug development business.
“We’ve forecast conservatively because we don’t want to over-promise. We want shareholders to make money. It’s still a stretch, don’t get me wrong, we’re tight on resources,” Mr Paterson told Stockhead.
“[But] If we find we’re [likely to miss the target], I’ll just switch off the expenses.”
A corporate review entitled ‘Code Red’ switched the business from a drug focus to device-seller.
Mr Paterson switched from chairman to CEO, cut funding to the drug business and refocused on the now flagship CardioCel technology used to correct heart defects and the under-development Transcatheter Aortic Valve Replacement (TAVR).
- Bookmark this link for small cap breaking news
- Discuss small cap news in our Facebook group
- Follow us on Facebook or Twitter
- Subscribe to our daily newsletter
He says the company was on “life support” when he took charge in March last year and was surviving on capital raisings.
“They were not that far from insolvency.”
Admedus (ASX:AHZ) has three business lines: bioscaffolds which include CardioCel and TAVR, infusion products such as syringes, and the immunotherapy division.
Analysts Stockhead spoke to were positive about the company’s fortunes — but they are keeping an eye on the company’s cash burn, the development of decent sales teams in the US and Europe, and the fate of the immunotherapy business.
Bargain basement stock
Shares in the medical device maker have been in the dungeon for years, with a spike between 2013-2015 when the CardioCel products began to hit the market.
On Tuesday Admedus closed down 4 per cent at 23.5c.
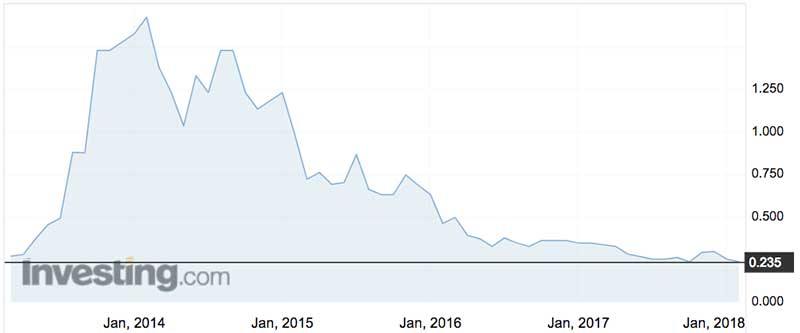
The company is not a takeover target because of that bargain basement share price.
That in turn means Admedus has to be careful about how they publicise the new TAVR device.
It’s a single tissue valve that is installed in the aortic artery via the leg, rather than via open heart surgery, and is competing with other products that involve more pieces of tissue and therefore more stitches.
Admedus wants to sign two of the big four global device makers to co-develop it this year.
But Mr Paterson says he can’t reveal whether it’s Medtronic, Edwards Lifescience, St Jude Medical or Boston Scientific in case they decide it’s simpler to buy the $60 million company while it’s cheap.
All four have TAVR programs.
Expansion plans
Last week Admedus launched the CardioCel product in India.
The company expects registration in China for early 2020 with South East Asia and Mexico next up.
Mr Paterson says they started the product distribution in the Middle East and North Africa because they had limited money, and he basically knew a guy in Lebanon who engineered the appropriate regulatory approvals in countries in the region.
They will start selling CardioCel in Egypt in the next few months.
Admedus is making money now.
In the December quarter it made $6.5 million in receipts, the most it’s ever made without having to rely on government tax refunds.
It still burned $3.4 million in the quarter — slightly less than in the September quarter.
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








