G Medical promises production is imminent as it gets $31m order from the US

Pic: Luis Alvarez / DigitalVision via Getty Images
Medical devices company G Medical is now promising that the production of its Prizma medical smartphone case is just around the corner a year after they ran into problems in China, and they have some news orders on the way.
It’s picked up up a $31 million purchase order fromUS healthcare business Hygea Holdings.
G Medical’s (ASX:GMV) Prizma case turns a smartphone into a clinical grade mobile medical monitor to measure vital signs such as the heart’s electrical activity (ECG), respiration, oxygen saturation, heart rate, temperature and stress analysis.
The US orders will commence at the start of April and cover two years. Payments will be made on a monthly basis.
Dr Yacov Geva said it placed the company in a “strong position” as it eyes off a listing on the NASDAQ.
It’s unclear how listing of their Chinese subsidiary in Hong Kong is going.
“Today marks our commercial entry into the key US market for our medically certified devices, and we are proud to partner with a well-respected and industry-acknowledged multinational of the calibre of Hygea,” he said.
China is now sorted
When we last checked in, G Medical had received the China Food and Drug Administration (CFDA) approval it needed to begin making its medical devices at its Guangzhou facility in China, months later than it initially hoped.
In an update posted to the ASX last week the company said device production would commence in “early 2019 once all final device componentry has been received for the production line”.
“The first units from the facility are expected to be delivered to customers in the first quarter of 2019 to territories which have further granted regulatory approval for the specific devices,” it said.
Sales to start?
Over the last two years G Medical lined up millions of dollars worth of sales contracts for the Prizma case, expecting these to generate revenue in 2018.
The China hold up, created by changes in the way the country regulated medical products, threw a spanner in this plan and created a cashflow headache.
- Subscribe to our daily newsletter
- Bookmark this link for small cap news
- Join our small cap Facebook group
- Follow us on Facebook or Twitter
G Medical has signed distribution deals with distributors SilverLake, Shandong Boletong and MEDTL since 2017.
The SilverLake deal for Chinese distribution was signed in May 2017 and didn’t specify a revenue figure, but did say a minimum of 4.05 million cases would be sold over five years.
The Shandong Boletong deal, signed in July 2017, was to distribute G Medical’s case and set up a medical call centre with support services was worth a minimum of $67.5 million in sales over 60 months.
And the MEDTL deal was to distribute in Cyprus and Greece and worth at least $US10.5 million in the first 12 months.
A $US405m distribution deal in Taiwan and India has stalled as the partner has so far failed to line up any buyers for the phone case.
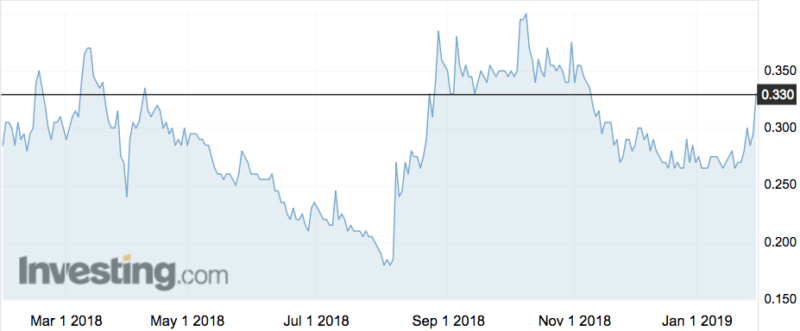
Shares rose 14 per cent on the news.
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








