Digital mental health assessment Medibio gets European approval

Pic: REB Images / Tetra images via Getty Images
Mental health tech play Medibio has won approval to sell its stress-testing products in Europe.
Medibio (ASX:MEB) was granted the Conformite Europeenene (CE) stamp earlier this month – certifying that the product complies with Euro health and safety standards.
“Acquiring CE Marking represents the successful completion of a lengthy and thorough evaluation process, and marks an important threshold for the company,” chief Jack Cosentino said.
“We are eager to progress to a new phase, and committed across the organisation to deliver exceptional product quality and operational excellence as we work towards transforming mental health care delivery.”
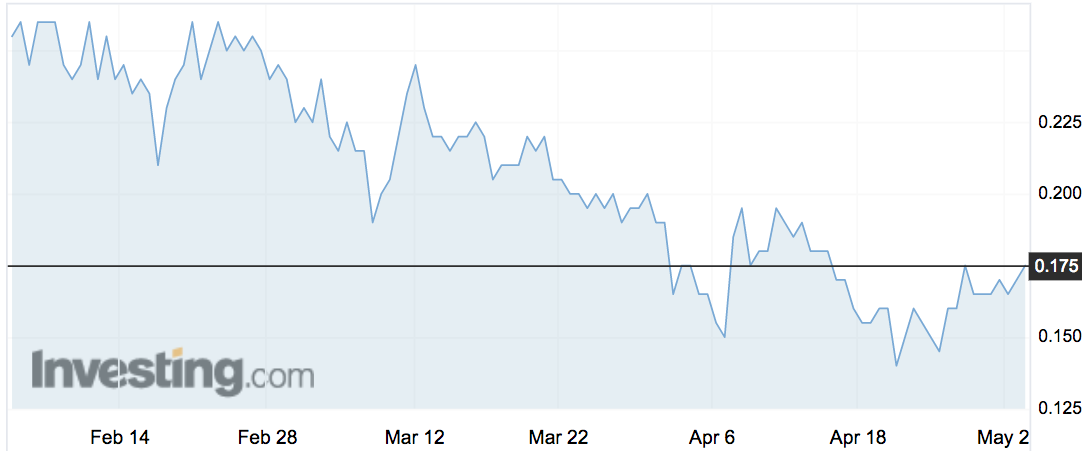
Shares in the company were trading up 3 per cent at 17.5c by 12.45pm AEST.
Medibio is pioneering the use of biometrics like heart rate and sleep statistics to assist in the screening, diagnosing, monitoring and management of depression and other mental health conditions.
Today’s news follows the launch of its corporate mental health program launch earlier this week.
The platform allows businesses and insurance companies to offer employees or customers the chance to “check-in on their mental health” using an app and to see results at an aggregate level to pick up data trends.
It says the product pricing is subject to firm size and structure, but initially ranges between $15 to $45 per user.
Medibio hopes to launch new applications for consumer health products — on devices from Garmin, Fitbit and Apple — in early 2019.
In the last quarter Medibio banked $61,000 in cash from customers and had $11 million in the bank.
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








