Zelda tries out weed to reduce opioid dependence

Pot stock Zelda Therapeutics has added another indication to the growing list of things it hopes the drug can treat: opioid addiction.
Zelda (ASX:ZLD) is starting clinical trials to “examine the potential to use certain cannabinoid medications in patients on chronic, high dose opioid pain management therapy”, alongside Melbourne’s St Vincent’s Hospital.
The company is already investigating the safety and efficacy of medicinal cannabis in treating conditions like insomnia, autism and cancer.
Prescription opioids such as oxycodone, fentanyl and codeine are used to treat chronic pain, but can cause dependence. The US is currently facing an opioid overdose crisis.
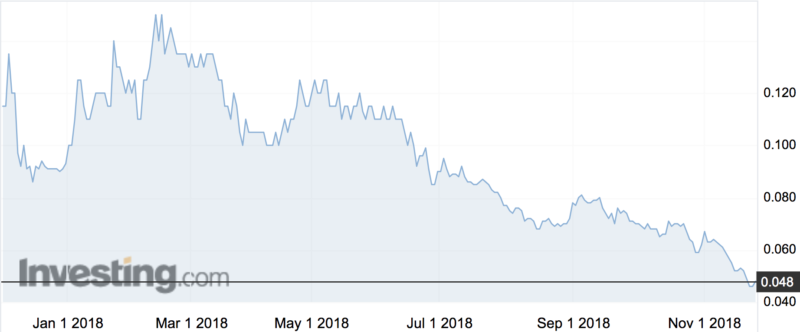
The company’s shares rose 7 per cent on the news to 4.9c, a much-needed boost as shares have been trading at year-long lows.
Zelda hopes that a joint clinical trial between itself and St Vincent’s will assist patients who have become dependent on opioids for chronic pain management, chief Dr Richard Hopkins says.
“Zelda is delighted to work with the team at St Vincent’s, which continues our partnership activities with world class institutes across the globe. While similar studies have been conducted in other countries with promising results, we believe that local medical community participation in these studies will accelerate acceptance of the potential of cannabinoid-based medicines,” he said.
An initial pharmacokinetic study will be 20 patient double-blinded, randomised, placebo-controlled study, with the potential to expand into a larger trial depending on results. Subject to approvals, the trial would be expected to start in early 2019.
Medication for the trial will be sourced from HAPA Medical, Zelda’s strategic partner for manufacture and supply of pharmaceutical-grade medicinal cannabis.
- Subscribe to our daily newsletter
- Bookmark this link forsmall cap news
- Join our small cap Facebook group
- Follow us on Facebook or Twitter
Zelda recently did a deal with former ASX tech wreck 1-Page.
It promised that company’s subsidiary HAPA Medical the first rights to distribute or make for the German market any formulations the biotech finalises.
Should the trial results show benefits then Zelda expects to be able to supply these medicines to Australian patients via specialists registered under the Authorised Prescriber Scheme.
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








