What’s next for Living Cell after failed clinical trial

Pic: sinology / Moment via Getty Images
At least Living Cell Technologies can take solace that it’s no Robinson Crusoe when it comes to failed clinical trials for treating the neurodegenerative condition Parkinson’s disease, which affects seven to 10 million people globally.
The late boxer Muhammad Ali and the extant Michael J. Fox are the foremost celebrity sufferers, with the time-travelling actor actively crusading for a cure.
Despite this advocacy no new Parkinson’s disease treatment has been approved in the US for at least two decades, with the standard-of-care drug levodapa now 50 years old.
The current treatments tackle only the symptoms, not the causes which centre on deprived levels of dopamine (a neurotransmitter that delivers messages to various parts of the grey matter).
Despite this limitation, Parkinson’s disease drug sales globally topped $US2.4 billion in 2014.
Auckland-based Living Cell (ASX:LCT) hasn’t altogether raised the white flag on developing its NTCell treatment, but the standard will be hoisted up the mast if further analysis of the trial results fails to come up with anything different.
What happened?
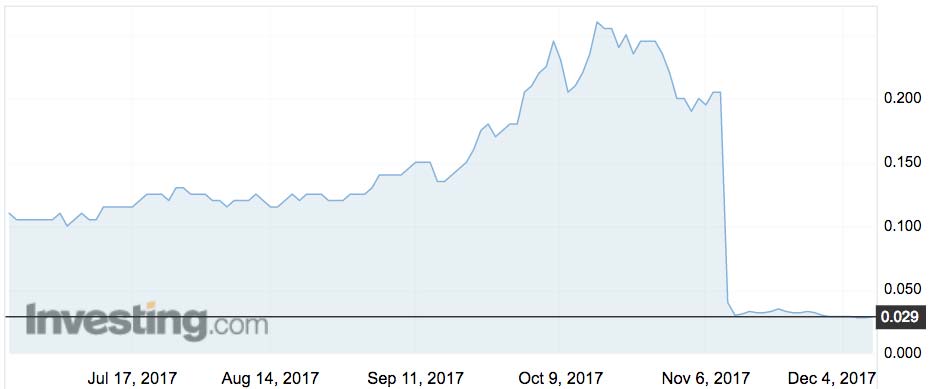
On November 10, Living Cell shares plunged by 85 per cent after the company reported “no statistical difference” between the efficacy of NTCell relative to the control group, in its 18-patient phase IIb trial.
The study was designed to confirm the most effective dose of NTCell, define any placebo component of the response and identify the initial target Parkinson’s disease patient sub-group.
The study consisted of three groups of six patients, with two patients from each group having sham surgery with no NTCell implanted. The active group received varying doses of NTCell implanted on each side of the brain.
While three of the four endpoints were met at the 26-week mark of the trial, they related to lack of adverse effects such as infection. But measured on the globally-accepted unified Parkinson’s disease rating scale (UPDRS), NTCell didn’t work compared to patients who underwent ‘sham’ surgery.
“It is encouraging that some efficacy data is positive and that the treatment was well tolerated and safe,” Dr Taylor said. “More data analysis and input from our advisors is required but at this time we cannot proceed with a regulatory application.”
He adds the company would not have carried out the trial any differently, such as choosing another endpoint.
“We did what we believed was the optimum study and we have to live with the outcome,” Dr Taylor says
Pigs might fly?
The NTCells are porcine encapsulated choroid plexus brain cells that provide neuroprotective factors and reduce toxins, derived from a herd of genetically isolated pigs from the even more isolated Auckland Island.
While not resiling from the fact the trial flopped, Living Cell has resolved to parse the data further, focusing on the patients that responded best.
Further 12 month results are due this month.
An earlier phase I/IIa trial resulted in the progression of Parkinson’s disease being halted for all four patients.
What’s next?
Living Cell had planned to seek rapid approval from New Zealand authorities, in view of a product launch in the first quarter of next year.
Suffice to say, the champagne order is on ice. The Living Cell board is meeting on December 20 and investors should expect a steer on what the company will do next.
Assuming the further trial results don’t result in any joy, Living Cell’s best hopes appear to hinge on developing the product platform for other neurodegenerative ailments, as well as retinal degeneration and chronic hearing loss.
Living Cell’s earlier prezzos and the company’s website also mentioned Alzheimer’s disease – another disease that has defied a cure – as well as Huntington’s disease and motor-neuron disease.
Earlier, Living Cell pursued a treatment for diabetes called Diabecell, also based on parts harvested from its special porkers.
The global rights to Diabecell are now housed in a US joint venture with Japan’s Otsuka Pharma Factory. The venture, Diatranz Otsuka, has licensed Otsuka Pharma to use Diabecell in the US and Japanese markets.
Past Parkinson’s flops
As Dr Taylor notes: “We are not the first company to get an encouraging first study placebo-controlled outcome and not meet the efficacy endpoint”.
One problem is that because there are variants of the condition, large patient pools are needed for a decent trial.
Last year a Bristol based phase II GDNF trial (as in glial cell-derived neurotrophic factors) failed to reach primary endpoints in an advanced trial. Similarly, a smaller precursor trial had produced “astounding” results.
Last month, shares in Acorda Therapeutics tumbled after revelations its $US380 million a year Parkinson’s disease drug Tozadenant could result in potentially fatal side effects including the loss of white blood cells.
Talk about the treatment being worse than the disease.
Another Innate?
Living Cell’s fall from grace mirrors that of fellow Kiwi based, ASX-listed biotech Innate Immunotherapeutics (ASX code IIL), which was backed by a bevy of White House heavies including Trump’s Congress go-between Chris Collins.
The Republican money men will think twice about acting on tips shared at the country club: Innate in June reported its advanced clinical trial for multiple sclerosis (another difficult neurological condition) had failed.
Innate confessed that while it would do follow-up analysis the results were unlikely to be different.
Innate has shut its operations and with residual cash of $4,335,000 is looking to buy another technology (or perhaps get into medical cannabis, lithium or Pilbara gold).
Sorry day
The Living Cell trial results came ahead of the company’s AGM, at which the apologetic board cancelled a proposed increase in the director remuneration pool, as well as a proposed issue of director options.
With Living Cell now valued at $17 million, compared with its cash backing of $6 million, investors expect the company to salvage at least some value from the clinical train wreck.
Diabecell, which is yet to be commercialised, may result in handy milestone payments of up to $15 million from Otsuka Pharma if development pans out successfully.
Dr Boreham’s diagnosis
We’re told the Auckland Island swine make for especially flavoursome bacon.
But whether Living Cell opts for charcuterie over alternative clinical work, the bald truth is that most shareholders were primarily interested in Living Cell’s Parkinson’s disease program, as they previously had been in the Diabecell cure for type 1 diabetes.
Founded in 1999 by University of Auckland paediatrics professor Robert Elliott and Kiwi company director David Collinson, Living Cell listed in August 2014 after raising $2 million at 20 cents a share.
The company has been around the block a few times and it’s understandable the statute of limitations on investors’ patience has expired.
If only they could borrow Michael J Fox’s DeLorean and take a spin through time to unwind their investment.
This column first appeared in Biotech Daily
Disclosure: Dr Boreham is not a qualified medical practitioner and does not possess a doctorate of any sort. His best White House tip was to put a lazy tenner on Trump at the last election.
This article does not constitute financial product advice. You should consider obtaining independent advice before making any financial decisions. The content of this article was not selected, modified or otherwise controlled by Stockhead. Stockhead has not provided, endorsed or otherwise assumed responsibility for any financial product advice contained in this article.
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








