What’s next for five-bagger Patrys on journey across the blood-brain barrier

Pic: Oscar Wong / Moment via Getty Images
Like the four-minute mile before fleet-of-foot medical student Roger Bannister cracked it in 1954, the blood-brain barrier has proved a stubborn obstacle for drug administration.
The brain is well-designed with a protective layer of cells allowing only certain molecules to transit from the blood to the cerebro-spinal fluid surrounding the noggin.
Is Patrys’ antibody cancer treatment PAT-DX1 one such molecule?
The Melbourne-based entity (ASX:PAB) seems to think so, hailing Yale University research showing the injected compound “crossed the blood-brain barrier to significantly reduce tumour size”.
Sir Roger, who died last month, would be proud.
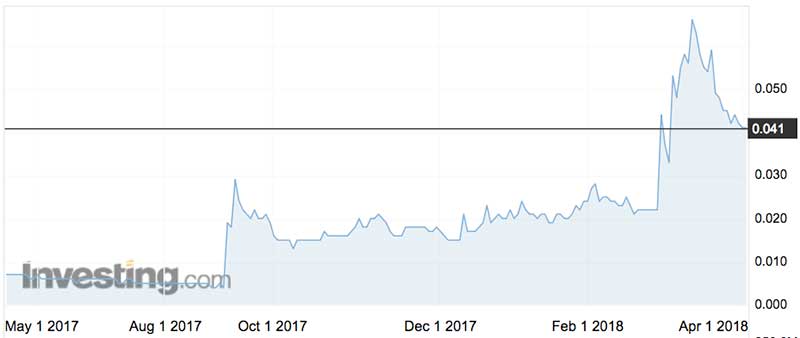
For investors it was a case of off-to-the-races as well, with Patrys shares more than doubling on the day.
“The blood-brain barrier is a really good layer of cells that stops bacteria, viruses and chemicals,” says Patrys chief Dr James Campbell.
“Few antibodies have managed to cross this barrier.”
Germanic roots
Patrys sounds like a Greek village or a 1970s orange juice brand.
In fact, the company was formed in December 2006 to consolidate human antibody technology from Germany’s University of Wurzburg, German biotech Oncomab and Acceptys Inc (a US company commercialising Columbia Uni know-how).
The company listed in July 2007, having raised $25 million at 40c apiece.

Patrys’ original work related to immunoglobin M (IgM) antibodies, which are bigger and more complex than the immunoglobin B antibodies used in most therapies.
In 2013, the company raised funds for a phase II trial, but it was curtailed because of manufacturing issues.
Dr Campbell, who has a background in biochemistry and venture capitalism, started as part-time chief executive officer in April 2015 and moved to full-time a year ago.
When he was appointed, the board asked Dr Campbell to see what he could do with the existing assets — but said he might also have to bring in something new.
Dr Campbell did just that, with the company licensing the relevant technology from Yale University in 2016.
A Chinese party has licensed the Chinese rights to a gastric cancer application for its old technology.
Otherwise, the old assets are on the books but dormant.
The lowdown on PAT-DX1
Patrys is a player in the art of DNA damage repair (DDR) therapeutics with a program called PAT-DX1.
PAT-DX1 is a humanised and smaller version of deoxymab 3E10 (D3E10), a DNA damage repair antibody first identified in the inflammatory immune disorder lupus.
While most antibodies bind to the surface of cells, D3E10 nanoparticles penetrate them and transport the agent across the protective plasma membrane. Rather like OMO on those tough stains.
It then binds to DNA and kills deficient or mutant cells.
The PAT-DX1 mechanism of action was “very different” to that of other antibody therapies, Dr Campbell said.
“This antibody is particularly cool because it localises in the tumour, crosses all membranes and binds to the DNA of the cell and stops the DNA repair enzymes.”
True, treatments known as poly-ADP-ribose polymerase (PARP) inhibitors block these enzymes as well.
But like OMO Plus, PAT-DX1 is the all-new-and-improved version.
In December, Patrys said it worked effectively with olaparib, the first approved PARP inhibitor.
In 2014, the US Food and Drug Administration approved Lynparza, Astrazeneca’s branded version of olaparib, for use in breast and ovarian cancer.
Patrys thus believes PAT-DX1 is likely to be used in other DDR therapeutics including with PARP inhibitors, as well as a vehicle to improve the delivery of chemotherapy and radiotherapy.
Multiple possibilities
PAT-DX1 has been shown to be effective in pre-clinical work on colon cancer cells and triple negative breast cancer (those lacking the three receptors that current drugs target).
In January, the company confirmed it worked in a similar localised manner for triple-negative breast cancers (the ones not showing the three usual markers).
Patrys also believes PAT-DX1 may be relevant for melanomas, prostate, pancreatic and ovarian cancers.
“We will go for glioblastoma first but there’s no reason not to think it will work in a broad range of other cancers,” Dr Campbell says.
The company hopes to start human trials within two years and has started the drudge work to enable this.
… but it’s early days
The Yale tests showed the glioblastomas — highly malignant cancers characterised by rapid cell reproduction — were 40 per cent smaller in the treated mice relative to the control rodents.
Putting the Yale results in perspective, they derived from orthotopic rodent models of glioblastoma using human tumour explants.
Under the orthotopic model, cells from human tumours are inserted in the brains of the mice.
Sceptics note that the tests pertain to mice and not men (with apologies to John Steinbeck there).
“There are models and there are models,” Dr Campbell says.
“People will say ‘what’s the point in curing cancer in mice’ but ours is a very sophisticated model.”
Yale’s parallel study to evaluate the comparative survival of mice with glioblastoma treated with PAT-DX1, relative to untreated mice, showed a 21 per cent survival increase.
Financials
At the end of December, Patrys’ coffers needed replenishing and a $2.4 million rights raising at 1.7c a share did just that.
The two-for-11 rights issue raised a net $2.4 million and was kindly underwritten by Somers & Partners.
As it happened the corporate financier didn’t need to do much for its fee, because the offer — struck at a 19 per cent discount — was 45 per cent over-subscribed.
Those investors were on the money.
Patrys shares doubled to 4.4c after February’s blood-brain breakthrough and last time we looked they were holding that level.
The stock has increased five-fold in the past six months. They have traded as high as 32c in March 2008 and as low as half a cent last August.
“I’m delighted the share price is starting to reflect the true value of the company,” Dr Campbell says.
Patrys investors include Mike Stork, of the Canadian tech investor Stork Holdings and Dr Dax Marcus Calder — a Perth periodontist with a passion for immunology who has supported the company since its early days.
Dr Boreham’s diagnosis
It’s well-known in big bad biotech land that only a fraction of early clinical programs get anywhere near commercialisation.
Thus, the $40 million market cap has to be seen as a funny money bet and we wouldn’t advise hanging around for a short-term dividend.
That said, the prawns are running M&A wise.
Late last year Johnson & Johnson bought antibody play Zymeworks for $US282 million, while in July 2016 Celgene acquired the early-stage Jounce for $US2.6 billion.
After Merck’s $500 million offer for our own mid-stage immuno-oncology play, Viralytics, anything can happen.
Disclosure: Dr Boreham is not a qualified medical practitioner and does not possess a doctorate of any sort. Given Patrys actually means ‘partridge’ in Afrikaans, he can only assume it’s a grouse company.
The content of this article was not selected, modified or otherwise controlled by Stockhead. Stockhead has not provided, endorsed or otherwise assumed responsibility for any financial product advice contained in this article.
This article first appeared in Biotech Daily
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








