Two medical device smallcaps are up, one is down after a soft quarterly

Pic: sinology / Moment via Getty Images
It was a busy reporting morning in the medical devices space Tuesday morning, with Osprey and Rhinomed posting positive December quarterly results while Volpara fell on missed guidance.
Osprey (ASX:OSP) shares were up 15 per cent to 15c on record quarterly sales of its flagship DyeVert product, aimed at making heart imaging procedures safer for patients with poor kidney function.
It says full-year unaudited revenue was US$2.5 million, up 54 per cent year-on year.
But even with today’s rise the company is still wallowing below a a not-very-well-supported capital raising priced at 15.5c in October last year.
And Rhinomed (ASX:RNO) jumped 8 per cent to 20.5c on news that the company made almost as much revenue in the first half of the 2019 financial year as it did for the total of 2018.
It posted $1.8 million in revenue, up 132 per cent year-on-year, and 82 per cent of the total revenue for the previous year.
The bad news
Volpara (ASX:VHT) is a medtech company trying to improve the early detection of breast cancer, through improved imaging techniques and advanced technology. The company makes money by licensing its software and imaging technology to practitioners.
It took record receipts from customers in the December quarter — their Q3 — pulling in $NZ1.9 million, a 192 per cent improvement on the same period in 2017, but missed its annual recurring revenue (ARR) and market share targets.
- Subscribe to our daily newsletter
- Bookmark this link for small cap news
- Join our small cap Facebook group
- Follow us on Facebook or Twitter
The company made $NZ5.6m in ARR in the December quarter.
But it had set itself an “aggressive” milestone of $NZ9 million by the end of the 2019 financial year, as well as servicing 9 per cent of US women with its software.
It now says it will reach “at least” $NZ6.6m, and reach 7 per cent of US women.
Volpara also has negative cash flow of NZ$2.8m, thanks largely to $NZ2.4 million in staff costs covering expansion of the US sales and New Zealand engineering teams.
Shares fell 15 per cent on the news to an intraday low of $1.05.
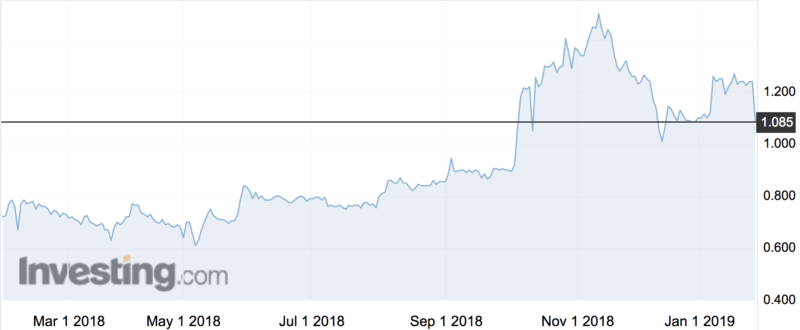
“We are pleased with our consistently increasing cash inflow and revenues and other key metrics, but we’re behind where we wanted to be at this point in the year for annual recurring revenue,” chief Ralph Highnam said of the results.
“The shortfall is due to a combination of the effects of the confusion caused by the introduction of GDPR [data laws] in Europe in mid-2018; needing to do trials outside the US, where we don’t yet have reference sites; our US sales team taking time to get fully productive; and the time it’s taking to close some of the very big US screening organisations.”
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








