Why Imugene has a fighting chance in its cancer battle

Pic: Luis Alvarez / DigitalVision via Getty Images
Earlier this year, Imugene managing director Leslie Chong asserted the cancer vaccine minnow did not want to be a “me too” company.
“We want to be innovative and come at it from a different angle,” she told a biotech confab.
As it happens, Imugene’s “different angle” was a $20 million licensing deal to acquire a family of 16 patents from the Ohio State University and Minnesota’s Mayo Clinic.
Hence, Imugene has swooped on the world’s biggest repository of B-cell patents and know-how.
The acquisition taps the 28 years of research expertise of Ohio University’s Prof Pravin Kaumaya, one of the godfathers of cancer vaccine research.
Ms Chong says Imugene cottoned on to the opportunity when chief technical officer Nick Ede chaired a conference president by Dr Kaumaya.
“We understood Prof Kaumaya was doing similar things but we didn’t know how far ahead he was,” she says.
Like a whirlwind romance, one thing led to the other and within seven weeks the Ohio/Mayo camp decided Imugene was its preferred partner.
The deal details
Overall the deal provides Imugene with six additional cancer targets.
To fund the shebang, Imugene is in the throes of a $20 million capital raising, by way of a $12 million placement and an $8.1 million rights issue, both at 2.7c a share.
Investors also receive a free option for every three shares held, exercisable at 4c each by November 2021.
The licence entails a small (six figure) upfront payment, milestone and royalty-style payments.
The raising is underwritten by Bell Potter so the funds are as good as through the door — although the deal requires shareholder assent at a meeting scheduled for July 9.
From poultry to immune-oncology
A classic Aussie battler, Imugene listed on the ASX in 2002, emerging from the shell of an outfit called Vostech.
Speaking of shells, Imugene initially was pushing vaccines for poultry (and pigs) and then turned its sights to a bird flu vaccine — but that opportunity flew away.
In 2012 Axel Hoos and Prof Ursula Weidermann identified the HER-vaxx technology at the Medical University of Vienna. The technology was incorporated as Biolife Science, which was initially private funded but then reverse-listed into Imugene (by then a shell company) in 2015.
Unusually, Dr Hoos remains on the Imugene board but he hasn’t given up his day job as vice president for oncology R&D at Glaxosmithkline (GSK).
Dr Hoos played a key role in developing the immune-oncology treatment Yervoy, aka the drug that saved Hawthorn AFL star and melanoma sufferer Jarryd Roughead.
Prof Weidermann is Imugene’s chief scientific officer.
Then Imugene’s chief operating officer, Ms Chong took over as CEO in 2016, replacing former Alchemia chief Charles Walker (who remains on the board).
In March, Ms Chong was elevated from CEO to managing director (in other words, she got a board seat but sadly no pay rise).
Ms Chong previously was senior clinical lead at Genentech, which developed the blockbuster breast and gastric cancer Herceptin before being acquired by Roche.
Chairman and gun negotiator Paul Hopper also chaired Viralytics, which Merck has just acquired for $502 million.
What’s the fuss about?
The Ohio-Mayo programs cover B-cell peptide vaccines and are closely linked to Imugene’s existing work. Its most advanced program is in phase II, covering solid tumours such as breast, colon and ovarian.
But the purchase also strengthens the prospect of developing so-called programmed death-1 (PD-1) checkpoint inhibitor drugs, in the same vein as the monoclonal antibodies Keytruda and Opdivo.
A combination therapy is also a likely outcome.
- Bookmark this link for small cap breaking news
- Discuss small cap news in our Facebook group
- Follow us on Facebook or Twitter
- Subscribe to our daily newsletter
Imugene’s lead molecule HER-Vaxx targets the same cancer receptors as the $US7 billion a year Herceptin – but Imugene hopes that it can do so more cheaply and with better efficacy.
HER-Vaxx is a mimotope: a small molecule, often a peptide, which mimics the structure of an epitope (the specific target the antibody binds to). The mimotopes cause the B-cells to produce a copy of the desired antibodies.
In non-nerdy terms, the vaccine wakes up the B-cells to produce millions of antibodies that fight the good fight.
The mimotopes work in the same way as Herceptin, but they are naturally produced and supposedly more efficacious.
Trial progress
Imugene’s story to date on the clinical front is a phase I breast cancer trial, completed at the Medical University of Vienna.
The trial enrolled 10 patients with late-stage HER-2+/++ cancers, in combination with traditional chemotherapy. HER-2 is a hair-like receptor found on the surface of 20 to 30 percent of breast and gastric cancer cells.
The results indicated a strong vaccine response, with the induced antibodies showing “potent anti-tumour activity”.
Attention has turned to a phase Ib/II trial for gastric cancers showing the HER-2+ mutation. The lead-in open label trial enrolled up to 18 patients, who were treated in combination with standard chemotherapy. Early patient data is expected this month.
(Ed’s note: Clinical trials are generally divided into three phases. Phase I focuses on safety, Phase II tests for effectiveness and Phase III examines whether the new drug is an improvement on existing treatment. Sometimes trials are further divided into parts A and B, where a B stage is generally more rigorous.)
A phase II study is expected to expand the patient cohort to 70 subjects across sites in Asia (the largest market for gastric cancer).
The Ohio-Mayo acquisition brings programs including a US National Cancer Institute funded, phase II HER-2 breast cancer trial, as well as a PD-1 checkpoint inhibitor for a phase I solid tumor trial.
The latter is described as ready for a US Food and Drug Administration investigational new drug application (IND).
The difference between the Imugene and Ohio-Mayo studies is that the latter carried out more academic investigator sponsored trials, while Imugene’s are structured more as commercial studies.
“Having parallel programs provides synergies and ergo that’s why we acquired [the assets],” Ms Chong says.
Financials and performance
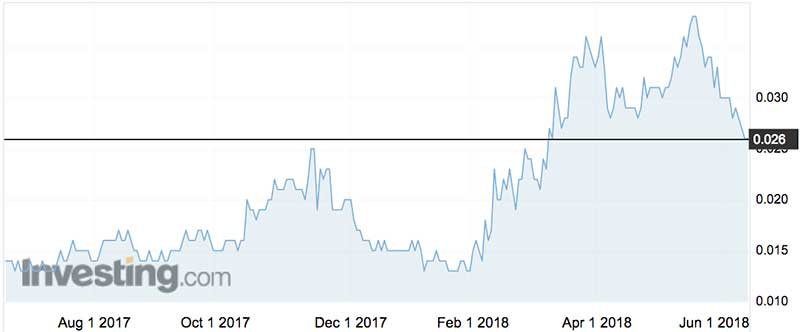
Imugene shares have traded between 1.2c and 3.6c over the last 12 months, having spurted from 1.9c to 2.5c after Merck swooped on Viralytics on February 21.
Late last year Imugene raised a chunky $8.7 million in a placement and rights issue.
Given the earlier raising was struck at 1.8c, it presumably delivered investors more than happy to pony up for the latest $20 million (which should be enough to fund the company for up to three years).
Imugene’s prospects were bright enough to attract Platinum Asset Management — which normally only invests in global companies — to the register.
It will be interesting to see what newbie investors crop up as a result of the raising, which will be completed next month following the shareholder vote.
Dr Boreham’s diagnosis:
Many biotechs have tried to develop cancer vaccines, but the Gardasils of the world (Prof Ian Fraser’s wonder cervical cancer vaccine) are few and far between.
With the company now tapping the world’s best B-cell brains, it has a fighting chance of getting somewhere.
Some pundits have questioned why Imugene is ASX-listed and with its HQ in Melbourne, given the development work to date has taken place in Johann Strauss’s old waltzing ground.
To date the company has regarded a Nasdaq listing as too expensive. For the time being, the company will remain as Australian as Vegemite (which unlike most of our supposedly dinky-di brands is actually locally owned).
“It’s a great time to be an Australian biotech, there are lots of things happening in the sector,” Ms Chong says.
Given the early nature of the combined programs it’s hard to say what Imugene should be worth, but it’s real value will become apparent as the programs either progress or stumble.
“If you follow the science, the dollars will come,” Ms Chong promises.
This column first appeared in Biotech Daily.
Disclosure: Dr Boreham is not a qualified medical practitioner and does not possess a doctorate of any sort. But he is as Australian as imported Holden cars.
The content of this article was not selected, modified or otherwise controlled by Stockhead. Stockhead has not provided, endorsed or otherwise assumed responsibility for any financial product advice contained in this article.
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








