Tim Boreham: What’s next for stem cell play Regeneus as it waits for Japan deal

Pic: sinology / Moment via Getty Images
With its early to mid-stage stem cell and cancer immunotherapy programs in the human and veterinary spheres, Regeneus has its fingers in some very hot pies.
But investors are waiting for something very specific: a pending licensing deal in Japan for Progenza, an off-the-shelf treatment for osteoarthritis pain.
On the company’s indications, they might not be waiting for much longer with a deal expected by early next year or even by Christmas.
Regeneus is already in rarefied company in Japan, having struck a 50-50 partnership with the country’s biggest drug manufacturer AGC.
The licensing partner would fund phase II trials and be responsible for distribution.
(Ed’s note: Clinical trials are generally divided into three phases where Phase I focuses on safety, Phase II tests for effectiveness and Phase III examines whether a new drug is an improvement on existing treatment.)
CEO John Martin says Regeneus was interested in five potential licensing partners, which was whittled to three, all of which are in due diligence.
In up-front monetary terms, he says the offers are similar. “It’s not a case of whose money is better but whether it will deliver a better outcome in terms of phase II and marketing.”
Eyes on Japan
Mr Martin says only 16 Australian companies to date have struck technology licensing deals with the Japanese, who usually are content to buy our coal and iron ore.
Regeneus will follow the path of stem cell leader Mesoblast, which already has an approved product in Japan in alliance with partner JCR: the graft-versus-host disease treatment Temcell, which it acquired from Osiris in 2014.
Cynata is partnered with Fujifilm and is seeking Japanese regulatory approval for its graft-versus-host disease treatment Cymerus.
Mr Martin says normally drug developers focus on the US, but Asia (notably Japan) is much more advanced. In the case of stem cells, the development push comes right from the top: Prime Minister and graft-versus-host disease sufferer Shinzo Abe.
“I don’t know of any major stem cell deal where the Japanese aren’t on the other side of it,” Mr Martin says. “Australian companies are well thought of in this space.”
The Regeneus evolution
The brainchild of prolific inventor Prof Graham Vesey, Regeneus was founded in 2007 by Prof Vesey and Dr Benjamin Ross, both of whom have Macquarie University links.
“Stem cells from adipose [or fat] tissue are not super novel, but he came up with a formulation of stem cells based on what they secrete and how they drive the therapeutic effect,” Mr Martin says.
The company listed in 2013 after raising $10 million at 25c a share.
Regeneus’s initial focus was on adipose stem cells in the veterinary sector and then an autologous human product called Hiqcell. (Autologous means the cells are derived from the patient’s own tissue.)
Being an autologous product, regulatory approval was not required and the company happily marketed the product for some years through a handful of Gold Coast and Sydney clinics.
But management then twigged that drug companies and clinicians were demanding allogeneic stem cell treatment; that is, using material derived from elsewhere and forming a more convenient off-the-shelf treatment.
Specifically, the company’s work centres on a sub-set of secretions called exosomes.
Mr Martin describes these agents as the “first responder at an accident site”, in that they give the body the signal to repair a cartilage, rather than being the direct regeneration agent.
Regeneus’s key products are Progenza and Sygenus, a topical dermatological treatment for acne.
The key difference is that Progenza consists of the cells and the secretions, while Sygenus contains the secretions only.
Does this matter? Well, sort of: Progenza is clearly classified it as a regenerative medicine while Sygenus is more of a biologic, which means a longer approval process.
All about Progenza
Technically speaking, Progenza cells excrete cytokines and growth factors to reduce inflammation and heal damaged and diseased tissue.
In its local phase I study called Step, the company enrolled 20 patients with moderate to severe knee pain.
In May 2017, the company reported the usual safety signals. But it also found a significant and “rapid and sustained” reduction in knee pain, as well as significant improvement in cartilage volumes and “positive signs of disease modification”.
In late 2016, Regeneus signed an exclusive deal with Asahi Glass (AGC), by which the maker of beer (and lots of other stuff) manufactures Progenza for the Japanese market.
Regeneus and AGC also formed a joint venture to develop and commercialise Progenza for osteoarthritis and all inflammatory indications in Japan.
The Japanese venture has established a cell production facility at AGC’s Yokohama Research Centre and it is developing processes for commercial manufacture.
Progenza has also been granted Advanced Therapy Medical Therapy (ATMT) classification in Europe. This recognises Progenza as a regenerative therapy within the EU.
… and Sygenus
Sygenus showed a strong analgesic effect in post-operative pain study, with longer lasting effects compared with morphine.
The study was the “first to demonstrate this effect from mesenchymal stem cells and their secretions”.
With acne, Sygenus in topical gel form showed reduced appearance of lesions after six weeks.
With age spots, 95 per cent were smaller and 63 per cent were smaller and lighter after eight weeks of treatment.
… and dogs
Our four-legged friends aren’t being ignored, either.
The company has a program called Cryoshot for canine osteoarthritis and Kvax, a canine cancer vaccine.
No, they’re not barking mad: these are huge markets.
Thus far, the company has treated more than 5,000 tail-waggers for osteoarthritis, and has not received a single written complaint.
“It’s a good program,” Mr Martin says.
“Not many companies have been able to get through a stem cell product for canine orthopaedics.”
“We have an established relationship with one of the biggest vet pharma companies in the world and we may have something to say about this over the next month or so.”
Finances and performance
Regeneus lost $5.18 million in 2017-18 compared with a $10.1 million profit previously. This sounds like an undesirable trend but the 2016-17 numbers reflected an $8.9 million milestone payment from AGC.
If the licensing deal is struck, another payment is due in the June quarter of 2019.
How much? Think in terms of a $US5 million payment but also a 50 per cent share of any upfront licence fees from a putative clinical partner.
At the end of September, Regeneus held $2.7 million in cash. The company has a $1.4 million loan facility from Sydney’s Paddington Street Finance, repayable at the next AGC milestone, the receipt of a 2018-19 R&D payment or by September 30, 2019.
Otherwise, the company hasn’t raised any equity funding in four and a half years.
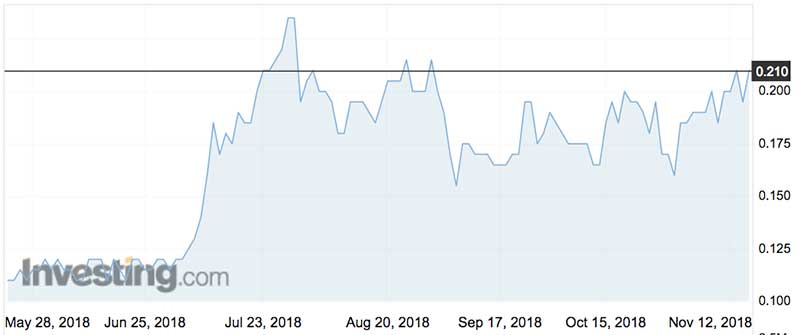
Over the last 12 months Regeneus shares have meandered between 10.5c in March this year and 23.5c in July. They peaked at 60c in November 2013.
Dr Boreham’s diagnosis
The proof of Regeneus’s success lies in sealing the vaunted Japanese deal and we always get nervous when boards nominate timelines for these transactions which are notoriously hard to execute.
As usual, we have the flattering comparison in terms of other global deals. In January, Takeda Pharma, Japan’s biggest pharma company, acquired Belgian regenerative medicine company Tigenix for a cool $US630 million.
The Regeneus board has been unusually stable and includes the five original directors including chairman and biotech legend Dr Roger Aston.
While the Aston-Martin combination drives the show, a notable addition to the board is veterinarian Dr Glen Richards, a judge on the Shark Tank commercial television show and founder of the listed vet clinic chain (and now takeover target) Greencross.
With the team racing for the finish line, it’s all eyes on the Japanese.
Disclosure: Dr Boreham is not a qualified medical practitioner and does not possess a doctorate of any sort. Like the dogs, he would love to have his belly fat removed and re-injected to make his knees, wrists and hips work properly again. The content of this article was not selected, modified or otherwise controlled by Stockhead. Stockhead has not provided, endorsed or otherwise assumed responsibility for any financial product advice contained in this article.
This column first appeared in Biotech Daily
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








