What’s next for high-flying dodgy knee fixer Paradigm Biopharma

Pic: Oscar Wong / Moment via Getty Images
It’s a case of everything old is new again at Paradigm Biopharmaceuticals, which is seeking to repurpose a very old drug for orthopaedic and viral arthritic indications.
Paradigm (ASX:PAR) certainly has amassed a new fan base and when it comes to spruiking the virtues of its anti-inflammatory lead compound pentosan polysulfate sodium (PPS).
Adelaide stockbroker David Baker speaks from personal experience.
A partner of Paradigm’s house broker Baker Young, Baker has used PPS injections to clear up his arthritis.
The drug is also widely used by 40 to 50 past and present AFL footballers to clear up knee and joint complaints and osteitis pubis, or groin inflammation.
Former Carlton and Adelaide star and Brownlow medalist Greg ‘Diesel’ Williams says PPS turned him from a virtual cripple to a re-energised guy able to go a full round of golf.
It had similar restorative effects on former Carlton high flyer (literally) Andrew Walker.
“He could hardly walk between practice sessions and had every available therapy thrown at him — but didn’t respond to any of it,” says Paradigm chief Paul Rennie.
“He is now running between 20 and 30km a week and has no knee pain and is playing [country] football again.”
Other than dodgy knees (osteoarthritis and bone marrow oedema lesions), current targets are viral arthritis, Ross River virus and Chikungunya (not a Sri Lankan village but like Ross River a viral infection spread by mosquitoes).
Phase I work relates to allergic rhinitis (yep, hay fever), chronic obstructive pulmonary disease and allergic asthma – proving that the plural of anecdote is not evidence.
Last year Paradigm’s phase IIa hay fever trial didn’t meet its primary endpoints, having touted that it could earn $1 billion a year.
(Ed: Clinical trials are generally divided into three phases. Phase I focuses on safety, Phase II tests for effectiveness and Phase III examines whether the new drug is an improvement on existing treatment. Sometimes trials are further divided into parts A and B, where a B stage is generally more rigorous).
The company blamed the formulation, so back to the drawing board on that one.
A new sense of purpose for old drug
Used in humans for more than 60 years, PPS is a semi-synthetic drug made from beech-wood hemicellulose.
Johnson & Johnson sells an oral formulation under the name Elmiron, to treat a painful bladder disease called interstitial cystitis. PPS is also used to treat deep vein thrombosis.
Paradigm secured a 20-year agreement Germany’s Bene Pharmachem, which makes the only US Food and Drug Administration-approved form of PPS.
Paradigm then listed in August 2015, having raised $8 million at 35c apiece in an oversubscribed raising.
Paradigm’s driving forces are 18 per cent shareholder Mr Rennie and chairman Graeme Kaufman.
Mr Rennie was Mesoblast’s head of product development. Mr Kaufman needs no introduction as former chairman of Bionomics and IDT, a former Cellmid director and continuing IDT director. He also was chief financial officer at CSL and an executive vice-president at Mesoblast.
Repurposing a drug means much lower development costs.
There’s also a 25 per cent chance of successful development compared with 10 per cent for a de-novo drug, with the development timeline abbreviated from 10 to 17 years to three to five years.
Paradigm intends to pursue the FDA’s 505(b) (2) pathway, by which the company can use historical data, in this case from Bene Pharmachem, to support the application.
About 30 per cent of approved new drugs are either reformulated drugs or combination drugs, so Paradigm’s approach is not especially unusual.
Paradigm also has the rights to a line of exosomes, which are bodies secreted by human cells and linked to the regeneration characteristics of stem cells.
Clinical progress
In all about 400 patients (mainly osteoarthritis sufferers) have been treated with PPS under the Therapeutic Goods Administration’s special access scheme.
The latest clump of results for 25 osteoarthritis patients reported a 60 per cent reduction in pain (as self-assessed by the patient) after six weeks of treatment.
Across 100 patients, the average pain reduction to date has been a little over 50 per cent.
Of course, this “real world” stories of broker’s dodgy joints and footy stars’ knackered knees does not amount to clinical evidence. Hence, the company has launched two key formal clinical trials for osteoarthritis and Ross River virus.
Paradigm has just finished recruiting 126 patients for the osteoarthritis trial, a randomised, double-blinded, placebo-controlled effort across six Australian sites.
The patients have concurrent bone marrow lesions, which result from fluid build-up in the bone marrow, which is generally the result of a ruptured anterior cruciate ligament or degenerative osteoarthritis.
First results from this trial are expected in the December quarter.
Paradigm has also recruited 20 patients for its phase IIa Ross River trial.
But last year its shares halved in value after the 80-patient phase IIa trial for allergic rhinitis failed to meet its primary endpoint.
Fast-track approval
Mr Rennie notes that for a 505 (b) application, the “real world” data can supplement the clinical data and enhances the chances of approval.
“Given the fact we are targeting chronic pain, the likelihood of fast-track approval is very high,” he says.
He hopes for a small pivot trial of as few as 100 patients, rather than the usual thousands.
The slimmed-down requirement raises the prospect of Paradigm funding the drug to approval stage, rather than finding a partner (which is always an option).
Initial results are expected this quarter for the Ross River trial and by the end of the calendar year for the orthopaedic trials.
Financial and performance
With a cash balance of around $4 million, Paradigm says it is adequately funded for the current trials, with potential capacity fund any phase III stuff.
Paragon received $955,317 from the exercise of in-the-money options during the quarter and then a further $464,285 in July.
It expects to bank a further $571,428 by the end of August.
The company also expects a $2 million Federal Research and Development Tax Incentive in September.
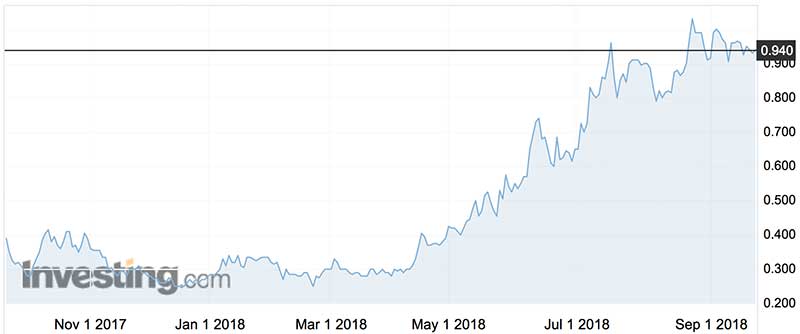
Paradigm shares have more than trebled over the last year and have traded as high as 96c in mid-July this year, and as low as 25c in August last year. The stock currently bears an $117 million market cap.
Dr Boreham’s diagnosis
Surely there’s no market for an effective remedy for dodgy knees and joints?
Just kidding!
The size of the global osteoarthritis market is estimated at $US5 billion a year, with 33 million creaky Americans alone suffering these ailments and a further three million here.
Mr Rennie points to a number of recent transactions that should spur investors. For example, in July last year Servier paid $US346 million for the European rights to Galapogas, an oral therapy for osteoarthritis.
If anything, Paradigm’s closest cousin is the Nasdaq-listed Flexion, which has had a drug approved for slow-release corticosteroids.
“It’s a good proxy for us in terms of market cap and clinical trial design,” Mr Rennie says.
While the ongoing results of the special access program are encouraging, investors will be holding their breath for the first phase IIb results by the end of this year.
“This will be a major inflexion point for us,” Mr Rennie says. “It will be one of the first clinical trials worldwide for a non-opioid, non-steroid based treatment.”
Of course, with all inflexion points the trial results have to inflex the right way.
Disclosure: Dr Boreham is not a qualified medical practitioner and does not possess a doctorate of any sort. He goes weak-kneed at the suggestion of a cure for dodgy joints.
The content of this article was not selected, modified or otherwise controlled by Stockhead. Stockhead has not provided, endorsed or otherwise assumed responsibility for any financial product advice contained in this article.
This column first appeared in Biotech Daily
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








