What’s next for Cardiex as ‘wearables’ trial gets underway

Pic: Oscar Wong / Moment via Getty Images
Adopting the philosophy of the (now late) Artist Formerly Known as Prince, Cardiex in June joined a swelling club of biotechs changing their name.
Think Avita (Clinical Cell Cultures), Immutep (Prima), Respiri (Isonea and Karmelsonix), Immuron (Anadis), Factor Therapies (Tissue Therapies) and Kazia (Novogen).
In the case of the cardiovascular health group Cardiex (ASX:CDX), the new moniker seems fair enough as it switches focus to the “wearables” and digital health sectors — markets with the claimed potential to increase sales 30-fold.
One aspect is a deal with the Silicon Valley wearable device outfit Blumio Inc, which could see Cardiex’s pulse wave analysis (PWA) diagnostic technique incorporated into Blumio devices.
Star studded board
Your columnist will dwell to an unusual extent on the impressive credentials of the Cardiex board, which in the main relate to the non-health sectors.
CEO Craig Cooper is a Kiwi-born, Australian-raised and now Californian-domiciled entrepreneur. Mr Cooper co-founded NRG Asia Pacific, Australia’s biggest independent energy producer.
A one-time Blake Dawson senior associate, Mr Cooper also founded Private Energy Partners and was CEO of software house E-world Technologies.
But wait, there’s more: in the US he co-founded Boost Mobile, was managing-partner of The Action Fund and has invested in new media including the Huffington Post and the irreverent Buzzfeed.
Mr Cooper is also co-host and investor in CBNC’s Adventure Capitalists, so is there anything this guy can’t do?
Chairman Donal O’Dwyer is (among other things) on the boards of Cochlear, Mesoblast and NIB Health.
Recent board addition Niall Cairns is a director of the ASX-listed Comops and Chantwest Holdings and managing partner of the Kestrel Growth Fund.
The US based Randall (or R as he prefers) King Nelson was prezzo and CEO of Uptake Medical, Kerberos Solutions and Venpro Medical.
Story to date
Atcor was born founded in 1994, based on the core Sphygmocor platform developed by co-founder Prof Michael O’Rourke.
Prof O’Rourke was a heart doc at Sydney’s St Vincent’s Hospital and professor of medicine at the University of New South Wales.
The company listed in November 2005 after a $15 million raising at 50c apiece. Co-founder Ross Harrick, a medical devices dude with experience at companies including Resmed, became inaugural CEO.
Marketed as Xcel, Sphygmocor is the only non-invasive, pulse wave analysis-based blood pressure device cleared by the US FDA as a 510(k) medical device.
This means the regulator deems it to be substantially equivalent to the current standard-of-care of an intra-aortic catheter pressure sensor insert.
The company claims Sphygmocor is the only device that can predict pre-hypertension and arterial stiffness, the main precursors to organ damage.
Effectively, the software takes a reading from the wrist or upper arm and measures central heart pressure and arterial stiffness.
“You might have great BP but your arterial stiffness might be off the charts,” Mr Cooper says.
Heart failure still accounts for 30 per cent of mortalities and snuffs out 20 million folk, annually.
To date, Cardiex has sold more than 4,400 Sphygmocor systems, mainly to pharma companies for research (the likes of Glaxosmithkline, Novartis and Astra Zeneca).
It also sells to hospitals (including the hospital affiliates of the Atlanta, Georgia-based Emory University and the Morrisville, North Carolina Sun Tech Medical) and has a licensing agreement with New York University.
Cardiex’s operations are based in Sydney and Illinois.
The new direction
Mr Cooper, who joined the company in early December 2017, says shareholders had tired of the company and many were disappointed when US reimbursement for the Sphygmocor Xcel was less than expected.
“My mandate is to turn the company around and refresh it,” he says.
A key focus of Cardiex’s new direction is on wearables and the blood pressure home-testing market.
With readmissions for BP tests clogging up hospital slots, health tsars understandably are keen to see more home testing.
Even now, seven of 10 Americans track health indications for themselves or a loved one — or their spouse — at home.
Cardiex’s new deal also entails a “direct to practitioner” push to a wider audience of GPs, executive health centres and naturopaths.
The company tugs at the purse strings rather than the heart strings by converting a “non-billable event” (brachial or standard upper arm cuff testing) into a billable one for these users.
Over time Cardiex has sold 4400 of the devices, which sell for between $10,000 and $17,000 apiece (a leasing alternative is available).
While that cost might be OK for the specialist cardiologists, it’s not for the wider target audience and the company needs to reduce unit costs to get the price of an Xcel unit down to around $5000 for the general (non-research) market.
“We can’t go into a naturopath like the way we walk into a specialist cardiologist,” Mr Cooper says.
While Cardiex’s current slant is on cardiovascular disease, it also plans to target obesity, orthopaedic health, diabetes and urology.
Jump-starting clinical trials
To kick off the wearables effort, Cardiex has announced a 15-patient trial, to be carried out at Macquarie University’s Faculty of Medicine and Health Sciences.
Over eight weeks, the Cardiex funded trial will assess the Blumio sensors’ responses to varying patient manoeuvres and the amount of cardiovascular data available.
“Basically, we are using our Sphygmocor technology as a predictive device alongside Blumio’s sensor for comparison of outputs in order to show that we can get acceptable blood pressure readings from the sensor,” Mr Cooper says.
The Macquarie effort is independent of Blumio’s US studies.
Financials and performance
Like a thirsty camel after a Sahara trek, Cardiex sure needed a decent drink with a December 31 cash balance of well under $1 million.
Luckily, the well — a placement and rights offer announced in April — was not a mirage and provided abundant water.
The “heavily oversubscribed” offer raised $2.37 million through a placement and $1.12 million from a rights issue (oversubscriptions of $907,882 had to be returned to shareholders). A related party, C2 Ventures has a binding commitment to chip in a further $1.5 million.
Both the placement and rights issue were done at 2c a share, a 23 per cent discount to the average one-month trading price and increased shares on issue by 89 per cent.
Cardiex has also acquired 7.5 per cent of Blumio for $US600,000 by way of convertible notes, with the first tranche of $US150,000 paid in June (the balance is due before the end of November).
Blumio is backed by investors including Peter Thiel, who co-founded Paypal and was an early investor in Facebook.
Not the guy you would want to un-friend.
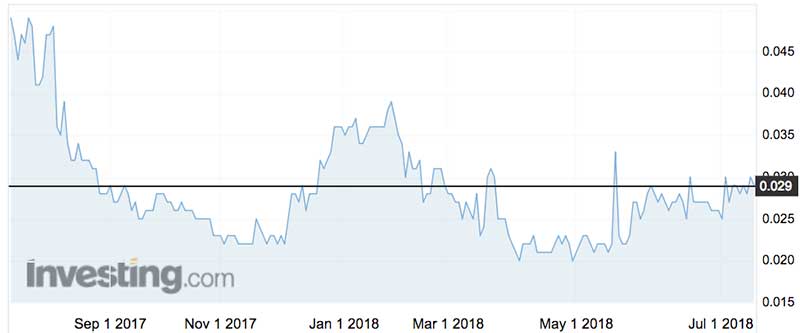
Over the last 12 months Cardiex/Atcor shares have traded as high as 4.9c (July last year) and as low as 2c (April 30 this year).
The shares peaked at 27c in October 2015.
Dr Boreham’s diagnosis
It’s understandable why management is entranced by the wearables sector, which is forecast to grow to $US8 billion by 2023.
The BP home testing space is also expected to grow to $US1.1 billion over this period.
With 2016-17 sales of $4.3 million — which management forecasts to be $4.5 million this financial year — Cardiex remains a modest concern despite its fancier name.
Take away the cash and the market is valuing the company at a mere $7 million, which Mr Cooper describes as “crazy”.
Cardiex is vaguely similar to Uscom (ASX:UCM), which is also in the central blood pressure measuring game (your columnist covered Uscom in February this year).
As well-credentialed as it is, Cardiex management still needs to prove its chops and get more revenue through the door.
This column first appeared in Biotech Daily
Disclosure: Dr Boreham is not a qualified medical practitioner and does not possess a doctorate of any sort. Any arterial stiffness belies a soft heart of gold.
The content of this article was not selected, modified or otherwise controlled by Stockhead. Stockhead has not provided, endorsed or otherwise assumed responsibility for any financial product advice contained in this article.
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








