Novita gets ready for US launch of attention deficit disorder tool

Pic: REB Images / Tetra images via Getty Images
Move over Thomas the Tank Engine and all aboard the Tali Train: a drug-free algorithmic-based tool to monitor and assist in treating cognitive disorders in children, notably attention deficit disorder and autism spectrum disorder.
Tali Train has already been available in Australia but in September the US Food and Drug Administration approved the tool as a class II medical device. This means it is exempt from the normal 510(k) registration requirements, notably further clinical trials.
In May, the Australian Register of Therapeutics Goods certified Tali Train as a class I device. Perhaps more importantly, the device is registered for what some cynics dub another (gravy) train: the National Disability Insurance Scheme.
Novita estimates about 400,000 Australian children, or 10 per cent of the juvenile populace, have attention deficit issues.
“In western countries, 136 million kids have been diagnosed with severe attention deficit disorder, so we figure there is a mighty opportunity for Tali Health,” CEO Glenn Smith says.
The company is eyeing a commercial launch in the US in 2019.
Owned by Novita subsidiary Tali Health, Tali Train is applicable for children three to eight.
https://www.youtube.com/watch?v=BiV7BR0-opw
Novita says the tool can be an adjunct to existing treatments, including psycho stimulants such as methylphenidate (best known as the brand name Ritalin).
Existing treatments (drugs) don’t treat the underlying causes and have side effects, with a question mark over long term impacts. They’re also not suitable for children under the age of five years.
Avexa hangs up its pick and shovel
Novita evolved from the old Avexa, which tried unsuccessfully to commercialize its HIV drug apricitabine before making a bizarre diversification into coal mining in Alabama.
The company returned home without even a banjo on its knee to show for its efforts.
In early 2016, Avexa acquired the Melbourne based Tali Health for $4 million, funded by a private placement. Avexa then carried out a 20-to-one share consolidation and changed its name to Novita in December 2016.
Avexa’s long serving chairman Iain Kirkwood departed in October 2017, while director Bruce Higgins bailed out in May this year.
Sue MacLeman joined the board in September, replacing Mark Simari as chairman (Mr Simari stays on as a director). Ms MacLeman is well known in biotech circles, including as chair of MTP Connect and Anatara Lifesciences, a director of Oventus and partner of Paul MacLeman, who once led Genetic Technologies and IDT Australia.
With a background in healthcare and recruitment, Glenn Smith was anointed CEO in October last year.
The Tali Train technology evolved from work by Monash University neuro-scientists and was developed in league with Torus Games and the commercialisation business (and Novita substantial shareholder) Grey Innovation.
Novita’s biggest shareholder, Peter Diamond, is the former principal of listed Perth stockbroker Euroz. He came on board more recently because he simply liked the story.
Former Carlton and West Coast champ Chris Judd also holds circa 5.7 per cent stake, through his investment company Three Zebras.
Now there’s a guy who never let his attention stray for four whole quarters.
Tali Train explained
Tali Train is an “adaptive fun-based interface” delivered under a software-as-a-service model. In effect, it’s a touch-screen game that adapts to the child’s performance and sharpens the challenges to increase attention skills.
The program involves 20-minute sessions five days a week, over five weeks. After that short period, clinicians will have a result.
Tali Train’s efficacy is supported by completed trials carried out at Monash University’s neuro-sciences division. Carried out over a long period, these trials showed Tali Train’s ability to focus kids on avoiding distractions while also honing academic skills (notably numeracy).
Mr Smith says the trial results have been supplemented by ongoing ‘real life’ data from the 150 clinicians on Tali Health’s books.
“Some of them effectively are in trials with us,” he says. “We do the same with pilot schools who are assisting us with performance data.”
Tali Health is also engaging directly with parents, whom the company hopes will become “Tali champions” and spread the word.
The company is also working on Tali Direct, a variation that can assess all children for potential problems. This variant is currently being trialed in some Victorian state and Catholic schools.
Recruitment sideline ‘fired’
Having acquired Tali Health, Avexa decided to accommodate both ends of the age spectrum by acquiring the Newly on-line recruitment site for $920,000 upfront, with an earn out of four times Newly’s 2018-’19 pre-tax profit.
Claiming one of the biggest databases of carers in Australia, Newly provides a platform for care or support professionals seeking work, or aged care or support providers seeking staff.
It then dawned on Novita’s management that there were no synergies between a kids’ digital health assessment tool and a jobs wrangler.
In July this year, a mere year after the purchase, Novita entered a heads of agreement to divest Newly to online market place Healthcarelink (HCL).
The deal involved Novita receiving HCL scrip equating to 10 percent of HCL, but with Novita contributing $400,000 to a HCL capital raising.
Financials and performance
In July, Novita raised $2.7 million in a placement to sophisticated investors and institutions, issuing 89.8 million shares at 3.1c apiece.
The company also garnered $3.5 million at 3c in July 2017 through a placement and rights raising.
The company’s inclusion on the National Disability Insurance Scheme (NDIS) approved list is crucial: of the 140,000 current scheme participants, about 38,000 (29 percent) have autism listed as their primary disability, while the portion is similar for those with an intellectual disability.
Of the autism cohort, 40 percent is in Tali’s target market.
Under the scheme, the company is charging $399 plus GST for each Tali Health package. The NDIS rules do not oblige providers to seek permission to set or increase their rates, but if their price looks like gouging they risk being chucked off the scheme.
Clinicians (usually paediatricians or psychologists) pay $299 but they’re encouraged to charge patients $399 to cover admin costs. Otherwise the clinicians benefit from more chargeable hours if the tests are done in situ.
The company expects Tali revenues to support cash flows “beyond the next 12 months”.
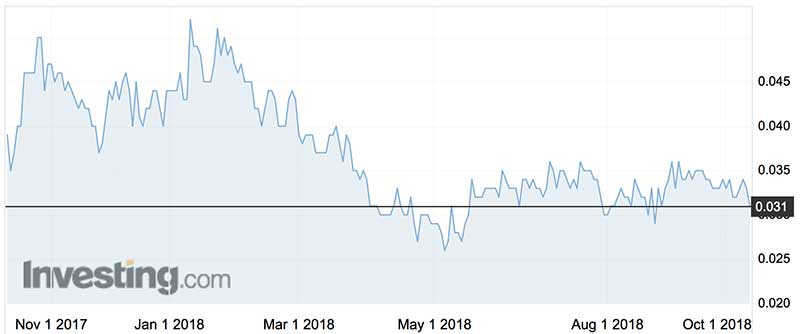
Under the Novita moniker, the company’s shares have traded in a range of 2.6 cents (December 2016) and 4.8 cents (January this year).
The current share price ascribes a lowly $14 million market capitalisation.
Dr Boreham’s diagnosis:
Relative to most other life science plays, there’s not much complexity attached to the Tali technology. But in a commercialisation sense, simplicity can be a wonderful thing.
Novita reports strong interest from overseas, with Tali Health setting up clinics in Singapore and an initial early learning centre in Hong Kong.
Competition wise, the coast is clear. A company called Akili recently raised $150 million to develop an assessment test for teenagers.
Mr Smith says it’s much harder to tackle the three to eight-year-old market because they are more difficult to recruit for trials.
“Other sophisticated university-based companies will seek venture funding to get in this early intervention space,” Mr Smith says. “We really have to hit this hard and get the key revenue indicators up and going.”
As a guide to the addressable market, Mr Smith estimates there are 500 relevant clinicians in Australia. If they apply Tali Train to one child every fortnight, that implies a $5 million a year opportunity.
Because the company started selling in earnest in July, the revenue uptick should become apparent in the September quarter results that are imminent.
Management warns that Tali Health’s commercialization strategy depends on factors including “assumptions relating to development and marketing expenditure, market demand, sales volume and pricing, working capital requirements and regulatory compliance”.
Okay! This may be stating the obvious, but it’s worth remembering nonetheless.
This column first appeared in Biotech Daily
Disclosure: Dr Boreham is not a qualified medical practitioner and does not possess a doctorate of any sort. His attention can wander off the tracks when Friday beer-o-clock looms.
The content of this article was not selected, modified or otherwise controlled by Stockhead. Stockhead has not provided, endorsed or otherwise assumed responsibility for any financial product advice contained in this article.
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








