How Invion plans to use light therapy to treat ovarian cancer

Pic: Charnchai / iStock / Getty Images Plus via Getty Images
In the crudest of summaries beloved of editors globally, Invion’s quest is about taking a treatment known for more than a century and improving it to tackle cancer.
There’s nothing about reinvention, of course: the machines that roll off BMW and Boeing assembly lines are also based on technology that was state-of-the-art at the turn of the century.
A few months ago, Invion shares were as unloved as a grey ghost hovering outside an emergency hospital.
The company had sought to commercialise its anti-inflammatory lead program INV102 (nadolol) as a smoking cessation aid — but could not interest any partners despite looking for two years.
“We were stuck in a hard place,” CEO Dr Greg Collier says. “The company’s market cap drifted below $10 million and the company went on ice for two years.”
A “virtual” company with three employees, Invion’s prospects were boosted by a key deal last year that injected a new asset: a light-based cancer treatment called Photosoft.
“We have plans in skin, ovarian and prostate cancers and other orphan indications, but our strategy is yet to be rolled out,” he says.
What’s the big deal?
The seminal transaction involved an exclusive distribution and licence agreement to commercialise and develop Cho’s Photosoft, a so-called photodynamic therapy (PDT).
Cho is the corporate vehicle of the Melbourne-based entrepreneur Michael Cho, who was aware of the common use of PDT in China.
Invion issued Cho Group $5.5 million in shares at 0.2 cents and Cho agreed to underwrite a $2.5 million rights issue at the same price.

This compact involved Mr Cho himself taking a 15 per cent Invion stake.
The clincher is that Cho Group funds all research and development and clinical trials for the cancer indications Invion will target.
In a separate tie-up, the local research institute Hudson Institute of Medical Research last month said it would partner with Invion to research and develop the next-generation PDT.
Hudson will provide research and clinical expertise, while Cho Group will continue to stump up the spendoolies.
Bonanza!
The Hudson deal sent Invion shares 50 per cent higher on the day — a bonanza for investors who subscribed to last month’s oversubscribed rights raising at 0.2 cents.
That raising was struck at a whopping 93 per cent discount to the prevailing price of 2.8c.
The twist here is that the raising was not meant to be at a discount at all, because the shares were trading at 0.2 cents at the time of the Cho announcement last February.
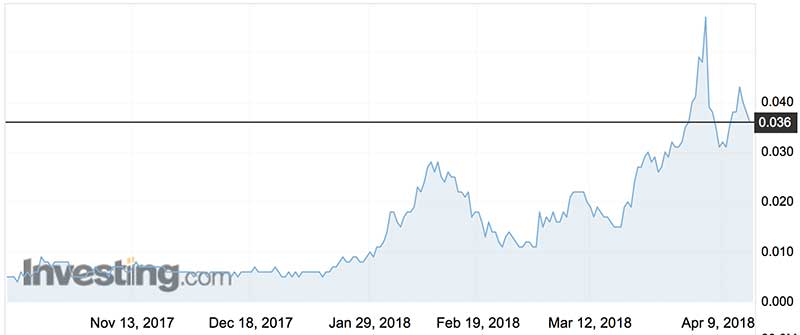
But interests associated with Cho just kept on buying, pushing the shares higher ahead of the Hudson announcement.
Given the discount on offer, punters queued to take up their rights and there was no shortfall for Cho Group to cover.
Holders had approved the terms of the raising at a meeting in November, so the company couldn’t adjust the terms of the offer to reflect the more lustrous share price.
History
Invion was formerly Cbio, which touted its XToll, or chaperonin 10, for inflammatory conditions such as rheumatoid arthritis and lupus.
In an ominous, but typically biotech signal, the company called a trading halt in July 2011 for the 155-patient, phase II trial results.
It then further semaphored surrender with a suspension to announce the trial very successfully demonstrated that the drug did not meet its primary endpoints, also known as it didn’t work. But it was safe.
A board spill followed, with legal action taken to recover $1.3 million from the previous regime.
In July 2012 CBio merged with Inverseon for its nadalol (INV102) for asthma and what it called “smoking cessation” and changed its name to Invion.
- Bookmark this link for small cap breaking news
- Discuss small cap news in our Facebook group
- Follow us on Facebook or Twitter
- Subscribe to our daily newsletter
Former Chemgenex chief operating officer (and now Patrys chief executive officer) Dr James Campbell was brought on board to repair the company.
In 2013, former Chemgenex chief executive officer Dr Greg Collier was given the hot seat, inheriting the Inverseon assets.
Trials showed that INV103 also didn’t work for lupus and INV102 increased abstinence and reduced the number of cigarettes consumed, which wasn’t quite the original claim of “smoking cessation”.
Cbio’s XToll, renamed INV103, is still on the books. So is INV102, but with Photosoft taking pole position it’s kind of been Photoshopped out of the picture.
Invion’s oncology bent is not surprising given that Dr Collier founded Chemgenex and eventually won US Food and Drug Administration regulatory approval for its chronic myeloid leukaemia drug Omapro, with Cephalon acquiring Chemgenex for $225 million in 2011.
Invion recently moved its HQ from Brisbane to Melbourne, making it much more convenient for the surf-loving Dr Collier to commute from his coastal Barwon Heads abode.
Photosoft explained
Photosoft uses non-toxic photosensitisers that react with visible light and oxygen to kill malignant cells in a chain reaction.
Photodynamic therapy (PDT) is thought to kill malignant cells, shut down tumours and stimulate the immune system. Photosoft uses a laser light activation method based on short, pulsating ‘near infrared’ (NIR) wavelengths.
PDT was developed by the Roswell Park Cancer Institute in the late 1970s. Roswell Park, just to clarify, is in Buffalo, New Jersey and not the New Mexico alien-spotting precinct.
PDTs have been used here to treat superficial skin cancers in a non-invasive manner.
Light therapy actually dates back to 1903, when Icelandic physiologist Niels Finsen snagged a Nobel Prize for his work in the area.
At least 500 trials have been carried out since.
In the 1990s it was used for prostate cancer and in 1995 the FDA approved the use of PDT for oesophageal cancer.
Last year Israel’s Steba Biotech won European approval for its light therapy prostate cancer treatment Tookad.
In the clinic
The Cho Group has funded two small prostate cancer trials in the past, which showed promising reduction in prostate size and stabilisation of prostate specific antigen (PSA) levels.
“We have plans in skin, ovarian and prostate cancers, as well as other orphan indications,” Dr Collier says. “But our strategy has not yet been rolled out.”
Under the tie-up with Hudson, Invion initially plans to focus on ovarian cancer, which kills about 1000 Australian women annually (with 1600 new cases every year).
Because ovarian cancer is normally detected too late, it’s hoped the treatment will reduce the incidence of ovaries removal.
Why Photosoft?
The so-called first generation PDT treatments were problematic in a few ways: they were derived from toxic materials and it took two to three months for the body to clear them.
These PDTs also had limited membrane penetration and thus efficacy.
Photosoft is chlorophyll (plant) based and only accumulates in the actual cancer cells.
It produces singlet oxygen, which is not a reference to the respiratory trends of Bali bogans in Bintang tops. Rather, singlet oxygen is a cytotoxic reactive agent that allows more effective zapping of the bad cells.
Dr Boreham’s diagnosis:
Invion’s billions of shares on issue remind everyone of its tortured history as Cbio, but one shouldn’t dwell on the past.
Dr Collier says the company has no need to raise more capital, so at least billions of shares won’t be added to.
As the only ASX-listed biotech in the PDT game, Invion at least has a rarity value.
Management has done well to snare the Cho and Hudson deals — the former involving the actual folding stuff — but it is very much a work in progress.
“We are at an early stage of restructuring Invion; there will be a lot more to come,” Dr Collier says.
The unanswered question is whether Photosoft has clear clinical advantages and much more work needs to be done.
Buffalo may be thousands of miles from New Mexico, but the truth is out there.
Cue spooky synthesised music.
This column first appeared in Biotech Daily.
Disclosure: Dr Boreham is not a qualified medical practitioner and does not possess a doctorate of any sort. He may have been abducted by aliens, but what’s normal anyway, these days?
The content of this article was not selected, modified or otherwise controlled by Stockhead. Stockhead has not provided, endorsed or otherwise assumed responsibility for any financial product advice contained in this article.
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








