How tech is disrupting the $2bn clinical trial market

Pic: Godji10 / iStock / Getty Images Plus via Getty Images
Special report: Health-tech company MedAdvisor (ASX:MDR) has announced how it is using its medication management platform to improve clinical trial recruitment.
One of the key challenges in clinical trials is patient recruitment. Clinical trial organisations and biotech companies are always on the look-out for new and innovative ways that can improve the speed to enrol patients to reduce the lead time between drug discovery and that drug being available to patients.
MedAdvisor is a leading medication management platform connected to over one million Australian’s. It has been able to use the valuable patient insights to connect eligible patients with selected clinical trials.
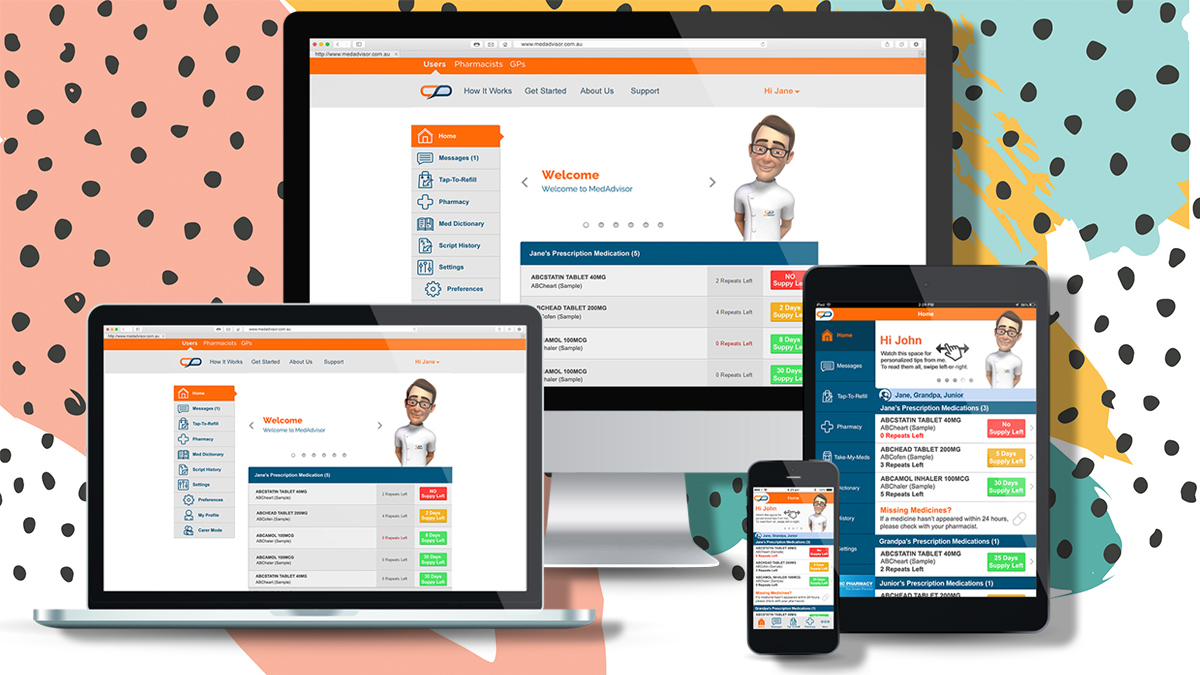
Clinical trials are not only recruited for based on the disease or ailment that a patient is affected by, but also by location, proximity to the trial centre and certain demographics. MedAdvisor has the ability to pre-screen all these factors and then connect eligible patients directly with the recruitment sites.
Growing market
Clinical trial recruitment is a $2bn industry and MedAdvisor isn’t the only firm to have identified this opportunity.
ASX listed biotech, Actinogen Medical (ASX:ACW), has partnered with dementia support app, MindMate, to help it drive patient enrolment into its Alzheimer’s disease clinical trial and the Federal Government have offered $12m in funding for medical research to make it easier for patients to find out about potentially lifesaving clinical trials.
ClinTrial Refer is an app in development to help connect patients with trials – something that MedAdvisor has been doing for some time now, albeit on a small scale that has allowed it to test and prove the functionality.
Success to date
To date, MedAdvisor has completed four trials and is actively offering its service to leading pharmaceutical and biotechnology companies. “There is no reason that we couldn’t roll out this service more broadly throughout Australia. We have a targeted approach, that can rapidly improve recruitment periods and improve processes for trial administrators,” said CEO, Mr Robert Read.
Read continued, “clinical trial recruitment comes with significant challenges in finding eligible patients in a cost effective and timely manner. 80% of trials are delayed due to patient recruitment and current recruitment methods can be very costly. We’re well positioned to leverage our growing database of patients to assist in improving patient recruitment methods and speeds and we have already successfully helped with a number of trials.”
Expanding its offering
The additional service adds to MedAdvisor’s existing offering and expands its revenue streams. Revenue is charged on a set up and per eligible patient fee basis. As the volume of trials increase, the opportunity to reach more patients to access new and innovative medicines improves.
MedAdvisor CEO, Robert Read said “We believe this is a win for patients and a win for innovative pharmaceutical and biotech companies who globally have struggled to find efficient methods of clinical trial recruitment.”
This special report is brought to you by MedAdvisor.
This advice has been prepared without taking into account your objectives, financial situation or needs. You should, therefore, consider the appropriateness of the advice, in light of your own objectives, financial situation or needs, before acting on the advice.
If this advice relates to the acquisition, or possible acquisition, of a particular financial product, the recipient should obtain a disclosure document, a Product Disclosure Statement or an offer document (PDS) relating to the product and consider the PDS before making any decision about whether to acquire the product.
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








