Dr Boreham’s Crucible: Does Osprey Medical’s GPO deal mean the cheque is in the US mail?

Pic: Oscar Wong / Moment via Getty Images
Having invented a device to reduce the usage of contrast dye used in x-rays, Osprey Medical (ASX: OSP) is heading to the GPO to spread the message that it’s well and truly open for business.
But we’re not talking about the General Post Office. For the Minnesota-based Osprey, GPO is an acronym for group purchasing organisation – powerful hospital-based procurement bodies that give the healing institutions greater buying power and help them to navigate reimbursement protocols.
In a breakthrough for Osprey’s US distribution, the company’s Dyevert products are being listed by four GPOs with coverage of half of all US hospitals.
These GPOs include the biggest, Premier, which has 4,000 member hospitals and channels $US50 billion of annual supply spend.
The Premier tie-up is a shift in focus for Osprey, which to date had been targeting heart specialists and hospitals one by one – a time-consuming way of doing things.
Be kind to kidneys
The reason for Osprey’s existence is that contrast dyes used in procedures such as stents and angiograms are toxic and thus hazardous to patients with vulnerable kidneys.
So much so, that the dyes induce a specific condition, contrast-inducted acute kidney injury (AKI). About 25 per cent of patients are considered vulnerable because they have poor kidney function in the first place.
A catheter-like device, Dyevert recovers a portion of the dye once it has done its job of guiding the cardiac surgeons.
Osprey then followed up with Dyevert Plus, an iPad-sized screen that monitors the amount of dye going into the patients and measures whether they have exceeded the recommended ‘dose’.
Under a printer and cartridge type revenue model, the ‘Plus’ components are virtually given away: Osprey makes its dough from the $US350 ($491) a pop, single-use Dyevert. Dyevert/Dyevert Plus is approved by both US and European regulators.
In the US, the Food and Drug Administration recognises Dyevert as the only product that can reduce dye usage.
The mail on the GPO strategy
Osprey has been selling directly in the US since 2015, starting with a sole sales rep in San Antonio but expanding to 27 operatives across 18 sales territories. Each territory covers 30 to 40 hospitals responsible for 35,000 to 50,000 heart procedures a year.
But it’s a hard slog winning doctor advocacy and then persuading the hospital to approve usage (which will only be for the specific surgeon initially).
The GPOs influence hospitals where it counts the most: the bottom line. In the case of Dyevert, that means reducing the cost of readmissions caused by patients contracting acute kidney injury.
Under US healthcare’s “patient outcome” based mantra – yes, we hate the term, too – hospitals are not reimbursed for ailments contracted as a result of the hospital visit.
In terms of the hard evidence, Premier kindly did much of the work for Osprey, by analysing the results of 2.8 million patients who underwent angiographs at 749 member hospitals between 2012 and 2017.
“Premier has the world’s biggest database of costs and complications and can compare hospitalisations,” Mr McCormick says.
Shockingly, AKI in the subset of acute patients adjudged to have bad kidneys increased from 18 per cent in 2012 to 28 per cent in 2017.
Premier assessed the financial burden at $US1.67 billion.
“They were stunned and we were stunned too,” Mr McCormick says. “That’s why we got a contract with Premier. They understood our value proposition because they looked at their own data.”
The reason for the increased kidney disease rate is rising global obesity and diabetes rates. Another is that angioplasty tools such as stents and wires are improving all the time, so more of these procedures are being carried out relative to bypass surgery.
McCormick says the GPOs don’t handle the goods themselves, but negotiate a contract price with a supplier. These supply terms are then sent to the hospitals, who continue to deal directly with the supplier.
“It’s not like Costco, the GPOs don’t stock and ship the supplies,” he says.
The Osprey guarantee
Strictly speaking, Osprey lacks formal clinical evidence of Dyevert’s efficacy, after a 2015 trial showed no reduced incidence of kidney disease. But the trial did show 15 per cent reduction in dye usage, with no reduction in x-ray image quality.
Along with adequate hydration via a drip and proper screening in the first place, the white coat brigade acknowledges reduced dye usage as a key factor in reducing kidney damage during a procedure.
To win over any doubters, Osprey is offering a money-back guarantee if the devices do not result in at least a cost neutral outcome for the hospitals.
Mr McCormick reckons he’s on safe ground: with each AKI event costing $US12,000, Dyevert only needs to reduce one such event in every 30 patients for the hospital to be ahead.
That adds up because using Premier’s negotiated discounted Dyevert price of $US333, the tool would cost just under $US10,000 for these 30 patients.
So far, Osprey has supplied 13,600 Dyevert units and has never had to repay a hospital. Mr McCormick says hospitals normally hate buying cutting-edge technology because the promised cost savings rarely materialise. And as the old maxim goes, no one gets sacked for choosing IBM (the proven safe provider).
“With Dyevert it’s easy to track the savings because the data is all there,” Mr McCormick says.
Financials and performance
Osprey posted a calendar 2018 loss of $US17.5 million ($24.5 million) up 22 per cent, despite revenue increasing 54 per cent to $US2.5 million ($3.5 million). Sales of Dyevert units increased 57 per cent to 7,201.
Osprey sits on cash of $US25.25 million at December 31, having bolstered its depleting coffers with a November 2018 capital raising that bought in $14.3 million. This included a $10 million placement to Allan Gray, which has emerged as a 14.9 per cent shareholder.
Osprey aims to be cash flow positive in late 2021 or early 2022, with the company focusing on the US business despite having the product approvals elsewhere.
“I don’t want to divert money into Europe right now,” he says.
However, Osprey is working on gaining approval from the UK National Institute of Health and Care Excellence, or NICE.
It’s not an easy task. But consent from NICE, which has access to similar acute kidney injury data to Premier, would open the way for Dyevert devices being available in UK National Health Service hospitals.
As for Australia, Osprey plans some pilot programs here, but it’s a hard market to justify given the Boston and New England market alone is bigger.
Since last July, Osprey has signed up 12 new US hospitals, 10 of them via the GPO process.
Osprey shares (Chess depositary instruments) listed in May 2012 at 40 cents. They were as high as 76 cents in August 2015 and are currently trading at record lows, around 13 cents.
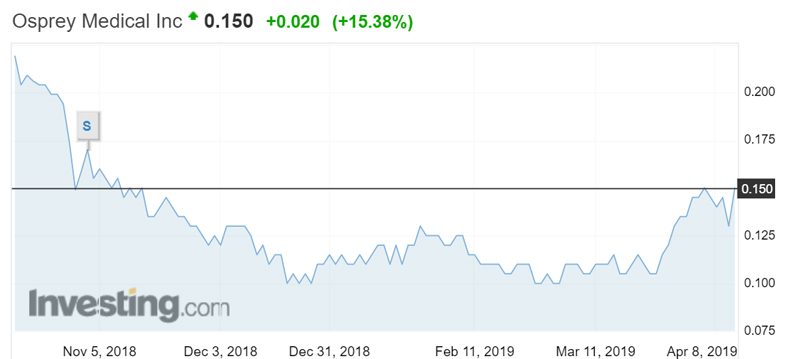
Mr McCormick says that given Osprey’s revenues doubled in 2017, investors may have assumed turnover would double again in 2018.
“That was disappointing for me and disappointing for the business,” he says. “I think it (the 57 per cent increase) should have been more robust.”
Dr Boreham’s diagnosis:
The Osprey board should expect a torrid time in relation to at least one agenda item at the company’s scheduled AGM in Melbourne on May 8. That’s not because the board isn’t doing its best to grow the company per se, but is lowering the performance incentive bar to the last stages of a limbo contest.
Just to explain: three directors were subject to several million performance options with a strike price of 30c, which is now in Darryl Kerrigan “you’re dreamin’” territory.
The solution? Re-issue the paper at Osprey’s current market valuation.
“The board believes that the outstanding out-of-the-money options are no longer effective as performance and retention incentives because they provide little or no value to option holders,” the company states in the notice of meeting.
But a hurdle price equivalent to the current lowly valuation? As Eildon’s most famous resident (at least before the fugitive Tony Mokbel) might say: “Fair suck of the sav, cobber.”
At last year’s Osprey jamboree, around 19 per cent of attending holders and 12 per cent of the total register opposed a similar motion to refresh the option arrangements for Mr Erb and Ms Lesenfants. As students of Australian corporate law would know, a 5.0 per cent holding is enough to call an EGM and initiate a board spill motion.
- Subscribe to our daily newsletter
- Join our small cap Facebook group
- Follow us on Facebook or Twitter
But let’s not get too Dyeverted by this dubious adjustment of the performance goal posts. The heartening aspect of the Premier deal for Osprey is that GPOs usually only deal with healthcare giants such as, say, Johnson & Johnson for billions of dollars of hip replacement parts.
A company with a $US3 million revenue run-rate would normally be told: “There’s the door and don’t let it hit you on the arse.”
Even though Osprey’s share price hasn’t exactly soared like its namesake bird of prey, Mr McCormick says the company is happy to remain exclusively ASX-listed.
“We’ve found the Australian market to be outstanding and we’ve never tried to raise funds in the US,” he says. “The knowledge of Australian investors about medical devices is very high and it’s been a good supportive capital market for Osprey.”
We’re confident that like ‘our’ GPO, Osprey will deliver. But like a household awaiting a second-class mail delivery, patience is required.
This column first appeared in Biotech Daily.
Disclosure: Dr Boreham is not a qualified medical practitioner and does not possess a doctorate of any sort. He’s learnt that GPO also stands for Good for
Parts Only, which pretty much sums up your columnist.
The content of this article was not selected, modified or otherwise controlled by Stockhead. Stockhead has not provided, endorsed or otherwise assumed responsibility for any financial product advice contained in this article.
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.