The ASX’s only gene therapist Benitec is undergoing clinical trials this year

Pic: Oscar Wong / Moment via Getty Images
Anyone who thinks that gene therapies are a biotech fad should heed the words of US Food and Drug Administration commissioner Scott Gottlieb, MD.
“I believe gene therapy will become a mainstay in treating and maybe curing many of our most devastating and intractable illnesses,” the good doc intoned recently.
“We’re at a turning point when it comes to this novel form of therapy.”
Globally, around 2500 gene therapy studies are taking place, including 120 phase II or III clinical trials.
In December, the FDA approved the first gene therapy: a drug called Luxturna to treat vision loss.
Enter Benitec (ASX:BLT), the only ASX-listed gene therapist and a rare exponent of “gene silencing”.
This has nothing to do with squeaky Levis, but is the art of turning off troublesome cells that cause diseases such as cancer. (Antisense is also involved in gene silencing, but Benitec’s science is made in Australia, while Antisense is a US import.)
Benitec’s know-how involves gene-silencing, or DNA-directed RNA interference (ddRNAi) to turn off the suspect genes.
The company’s “silence and replace” credo sounds like something that happens to out-of-favour members of the North Korean politburo.
But it actually involves the process of silencing the mutant gene while adding a copy of the normal version of the gene.
How cool is that?
“When people crack it, it will be the big deal,’’ says Benitec chief executive officer Greg West.
Wrong way, go back
Not that Benitec’s evolution has been straightforward, with the company abandoning its original hepatitis B program with California’s City of Hope hospital and then going silent on a China hep B program, as well as a small-cell lung cancer tilt before abandoning its make or break TT-034 hepatitis C treatment, despite backing from Pfizer.
In a variation on Kerry Packer’s “only one Alan Bond in a life-time”, Pfizer spent a shirtload on the drug but handed it back to Benitec because other hep C drugs had been developed in the meantime.
But Pfizer’s investment (rumored to be $30 million) won’t be wasted, as it has produced valuable data for Benitec’s hep B program.
A series of US patent challenges delayed progress for years, but eventually Benitec and the CSIRO won the day for the Graham ’099 patent, which is the basis of all its gene silencing work.
Founding scientist and inventor Dr Michael Graham remains on the Benitec team as head of discovery
At least Benitec has stuck to its remit since listing 16 years ago, on the gung-ho promise of “unleashing a global storm’’ in gene silencing.
And over the years, Benitec has had a who’s who of Australian biotech players, famously swapping CEO Sue MacLeman for Progen’s then Dr John Chiplin and Dr Mel Bridges in 2010.
Other bacon, lettuce and tomato (BLT) alumni include New South Wales lawyer Ray Whitten, Novogen’s (now Kazia) Iain Ross, as well as Fermiscan and Bioxyne’s Dr Peter French, Sienna’s Dr Cliff Holloway along with Sakura Holloway, Analytica’s Carl Stubbings and Analytica and Viralytics’ Bryan Dulhunty.
The DNA of Benitec
Benitec the listed entity was formed from the shell of Queensland Opals NL (as in No Luck) and debuted on the ASX in July 2002, having raised $19 million at 50c apiece.
Benitec listed on the Nasdaq in 2015. Last year it filed a US shelf registration, which streamlines the US fund-raising process.
As with so many biotechs, Benitec’s roots trace back to the venerable CSIRO, which hived off the technology pre-listing and still owns a minute stake in Benitec.
In 2011 Benitec changed its name from Benitec to Benitec Biopharma to “better reflect the company’s current activities” and the world kept turning.
Get with the programs
Benitec’s two lead programs are undergoing clinical trials this year.
The first, BB-301 tackles oculopharyngal muscular dystrophy (OPMD), a rare genetic condition that affects about one person in every 100,000 (12,000 western world sufferers).
OPMD is characterised by eye-lid drooping, difficulty in swallowing and limb weakness, with the sufferers likely to die from malnutrition or pneumonia.
The BB-301 program is designed to treat the dysphagia, the difficulty in swallowing associated with the disease.
But it is hoped that the single-injection treatment might restore muscle tissue and — dare we say — cure the disease.
Last November, Benitec applied to the FDA for orphan drug designation for BB-301, which affects fewer than 200,000 Americans.
The company already has orphan status for OPMD in Europe.
The OPMD trial will involve 20 to 30 patients being injected weekly into a throat muscle, over eight weeks.
The trial locations are yet to be decided, but here’s a clue: OPMD is a “cluster” disease prevalent in France, French-speaking Canada, New Mexico and Israel.
Traditionally, the standard measure of swallowing is the time it takes the patient to down a glass of water.
But Benitec is looking at more quantitative measures, including real time x-ray imaging.
The second advanced program, BB-401 tackles head, neck and shoulder cancer (HNSCC), which Mr West describes as a “debilitating and hard to treat disease associated with a poor prognosis”.
BB-401 targets a protein called the epidermal growth factor receptor (EGFR), which is over-expressed when tumours are present.
About 64,000 new HNSCC cases are detected in the US alone annually, with 13,000 deaths. The EGFR protein is over expressed in about 90 percent of HNSCC cases.
This program was bought to the table by Nantworks, a company owned by billionaire Patrick Soon-Shiong, said to be the world’s wealthiest doctor, ahead of even American telly’s Dr Phil.
A related entity, Nant Capital, owns a chunky 29 per cent of Benitec.
The BB-401 open-label trial intends to recruit up to 30 patients who have failed all standard therapies, across five to eight sites in Australia and Russia (where smoking and drinking rates are high).
The primary endpoint? Reduced tumour size or complete ablation.
Financials and performance
The “global storm” of gene silencing promised all those years ago has been more a case of shifting breezes with plenty of headwinds.
Still, Benitec is well placed with around $10 million in cash. And with the backing of Nant, never say you can’t.
With HNSCC, Benitec is vying for a drug market currently worth $386 million across the western countries that count, with the market forecast to grow to $1.53 billion by 2024.
Benitec also pocketed a $4.11 million Federal Government R&D Tax Incentive fund in January and received a total of $10.5 million in grant income in 2016-17.
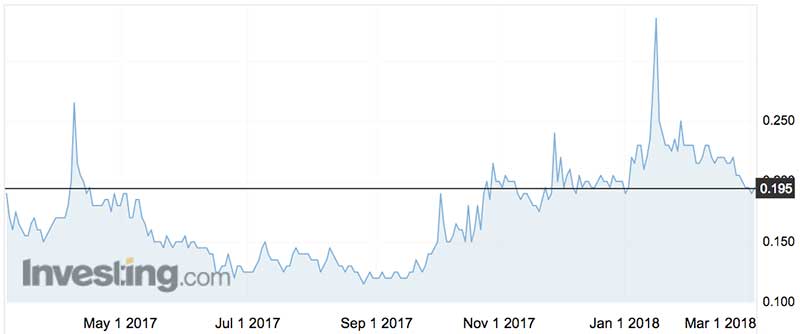
The shares are prone to some crazy-ape moves, having blipped like an errant cardiogram on three occasions in the last year.
On October 19, 2017, the shares closed 20 per cent higher on the soporific news of a US patent grant in relation to hepatitis B.
Over four trading days in January this year the stock spurted 60 per cent to a two-year high of 33.5 cents, around news of the orphan designation for BB-301 (the OPMD stuff).
What to expect
Benitec’s a fun stock to trade, especially in the lead-up to key events.
Seeing that you asked, these include the current-quarter launch of the HNSCC phase II confirmatory trial.
The OPMD program moves from lab to clinic by the end of 2018, with retinal disorders and infectious disease programs following in 2019.
In the case of the hepatitis B BB-103 program, work to date has been on mice with “humanised” livers (which does not mean they were force fed 10 Martinis a day).
Having met with the FDA, Benitec reports a “clear and expeditious path to clinic’’.
But Mr West says the company plans to partner out the program and has had some deep and meaningful discussions already.
Benitec’s other programs for retinal disorders and infectious diseases “could be” clinic-ready in 2019.
Benitec’s programs relating to HIV/AIDS cancer immunology, Huntington’s disease and intractable neuropathic pain have been licensed out.
Dr Boreham’s diagnosis
Unlike Scott Gottlieb MD, your humble faux practitioner finds the science around this one especially dense.
Or maybe he’s just dense.
Benitec chief scientific officer Dr David Suhy does a sterling job explaining it (with lots of diagrams) in a preso to the Biotech Showcase on January 8.
Interestingly, the Benitec register has been populated by doctors (and not just billionaire ones) putting in their own dough. So they get it, too.
“The initiation of the two clinical studies will be the major catalysts for value creation and news flow in 2018,” Mr West says.
Let’s hope at least one of the results puts a rocket up the share price — a concept any dumbo could understand.
This column first appeared in Biotech Daily
Disclosure: Dr Boreham is not a qualified medical practitioner and does not possess a doctorate of any sort. He failed the Densa test – which is actually quite hard – with flying colours.
The content of this article was not selected, modified or otherwise controlled by Stockhead. Stockhead has not provided, endorsed or otherwise assumed responsibility for any financial product advice contained in this article.
This article does not constitute financial product advice. You should consider obtaining independent advice before making any financial decisions.
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








