As health tech soars, these two ASX medical device plays might have flown under the radar
EMV and MVP are two of the emerging ASX health stocks on the ASX. Picture: Getty Images
With health issues at the global forefront over the past year, it’s no surprise that Australia’s health-related sector has also grown substantially.
But the industry was already emerging even before COVID-19. Excellent research facilities and a flexible regulatory regime has made Australia one of the leading players in biotech and pharmaceutical innovation.
According to Austrade, Australia ranks very strongly globally for its competitiveness in biotechnology and as a location for clinical trials.
And with so many health companies emerging in the ASX, it’s easy to miss who’s doing what.
Stockhead has the latest compilation of health and biotech related stories to keep you abreast of developments.
In this article, we will focus on two ASX medical companies that might have flown under the radar – EMVision Medical Devices (ASX:EMV), and Medical Developments International (ASX:MVP).
Both companies have cutting edge products that are sold in global markets, and are set to grow significantly over the coming years.
EMVision (ASX:EMV)
EMV focuses on medical imaging technology used in detecting stroke, and aims to disrupt the stroke care industry in a world where two-thirds does not have access to diagnostic imaging.
Rather than taking the patient to the medical imaging device however, it wants to bring the imaging to the patient by using its portable, electromagnetic RF imaging device technology.
The company says that transporting patients from ICU to radiology can increase the risks of complications, with adverse events occurring in as many as 71 per cent of in-hospital transports.
Logistical challenges in transporting unwell patients include moving critical but cumbersome devices like monitors and oxygen tanks, which requires multiple hospital staff members to get involved.
In many instances, by the time the patient is transported to the radiology department, it may be too late.
EMV says its aim is not to replace the CT or MRI, but rather to function much like ultrasound does, by providing clinically valuable information to healthcare workers, wherever the patient is.
This allows healthcare workers to intervene and make critical decisions earlier, when time is of the most critical.
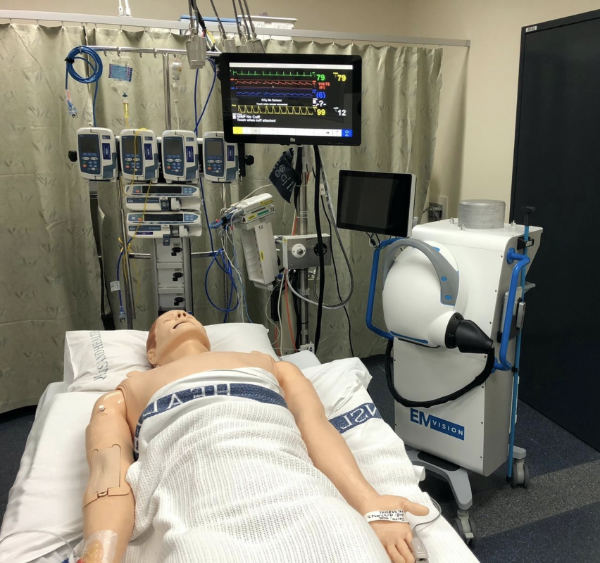
The product is the result of over a decade of research and development at the University of Queensland, where early prototypes were made.
A clinical trial is currently ongoing at the Princess Alexandra Hospital in Brisbane. The latest trial result in October 2020 showed the EMV imaging device was able to classify stroke type (haemorrhagic or ischaemic) with an overall accuracy of between 93.3% and 96%.
The study also concluded that the EMV device was able to localise targets in the correct quadrant, compared to CT/MRI, with an overall accuracy of between 86.7% and 96%. It has engaged with the US FDA as part of this trial.
The company has recently received an $8 million cash grant from the Australian Stroke Alliance, which will support the development of its planned first responder model for air and road ambulances, as well as its new brain scanner device.
The company is yet to make a profit, and its latest half year showed that it made a loss of $3.2 milliion.
The EMV share price, however, has risen by 16 per cent in the past month, and is up a massive 265 per cent in the last 12 months.
EMV share price chart:
Medical Developments International (ASX:MVP)
MVP is a medical company focusing on emergency pain relief and respiratory products.
Its flagship is the Penthrox drug, a non-opioid pain reliever which is self-administered in patients with trauma, and those requiring analgesia for surgical procedures.
The product is used in Australian hospitals, including emergency departments and ambulance services, as well as by the Defence force.
It’s also used for analgesia during short surgical procedures such as dental and cosmetic surgery.
Penthrox has also been approved in over 40 countries, but the uptake in Europe has been slow compared to Australia.
Ireland currently represents its biggest market in Europe, but still lags Australia by a long shot.
MVP has said that its immediate near-term objective is to increase its market share in Europe. The company is currently raising a total of $30 million in placements and SPP to accelerate the commercialisation of Penthrox in the EU.
It has also recently brought in a new CEO and non-executive chairman to push this agenda.
But the company is not just about Penthrox.
It also manufactures high-quality respiratory devices used in asthma care. The latest product in this segment is the Space Chamber, which can be used with an asthma puffer to boost and deliver more medication directly into the lungs.
In the latest half year, MVP’s net revenues increased by 15 per cent to $12.5 million, for a net loss of $1.1 million, saying that it was a reflection of the many challenges presented by COVID-19.
However, it announced a bullish outlook and expects a brighter year ahead – as it grows market share for Penthrox in Europe, and its respiratory franchise in both Europe and the US.
MVP share price chart:
Related Topics

UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








