Argenica’s latest HIE trial results show prolonged reduction in brain injury

The company is targeting an investigational new drug application with the US FDA. Pic: via Getty Images.
Biotechnology player Argenica Therapeutics has announced the latest positive preclinical data in hypoxic ischaemic encephalopathy (HIE) – which is when not enough oxygen or blood goes to a baby’s brain causing brain injury – with a single dose of the company’s ARG-007 drug has been shown to last out to four weeks.
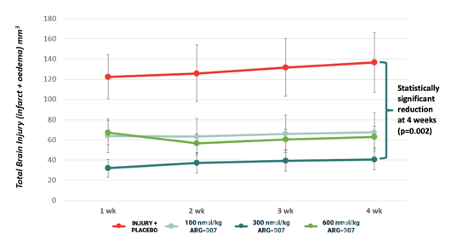
ARG-007 has been shown to significantly reduce brain injury caused by both ischemia (reduced blood flow) and vasogenic oedema (brain swelling) in a full-term-equivalent animal model of HIE.
A 300nmol/kg dose of ARG-007 maintained over a 70% reduction in total brain injury from one week out to four weeks post injury and doses of 100 and 300 nmol/kg of ARG-007 also reduced vasogenic oedema (brain swelling) at 48 hours post injury by 35.4% and 32.9% respectively.
The prolonged effect of the drug is particularly important as HIE is an ongoing process with three waves of neuronal cell death occurring over hours to days after the initial event, so a neuroprotective drug must have a prolonged effect to give the brain the best chance of protection against the third wave of cell death.
A solid base to move into clinical trials
Argenica Therapeutics (ASX:AGN) is now in the process of completing preclinical studies required to initiate an Investigational New Drug Application with the US Food and Drug Administration (FDA).
MD Dr Liz Dallimore said the preclinical efficacy data is “extremely encouraging.”
“Whilst HIE is a rare paediatric condition, it has devastating outcomes for these babies, and a treatment is desperately needed,” she said.
“The fact that ARG-007’s effect in this preclinical model lasts out to four weeks indicates the treatment effect is sustained, protecting against the devastating third wave of brain cell death seen in HIE.
“This data, plus the additional data we will gather in larger animal studies, will provide a solid base to move into clinical trials for HIE.”
Next trial phase planned in the US
AGN has engaged global contract research organisation Labcorp Drug Development’s (Labcorp) paediatric regulatory team to develop a regulatory and clinical trial strategy for ARG-007 in HIE in newborns.
And to meet requirements to undertake clinical trials in HIE in the US, AGN has initiated a preclinical juvenile toxicology study and preclinical efficacy studies in a large animal term model of HIE.
If these studies elicit positive results, then the company’s aim is to commence a Phase 1/2 trial in HIE in the US.
The results of the preclinical efficacy studies (funded by a grant from the Stan Perron Charitable Foundation) as well as the company’s engagement with the FDA, will be announced as they come to hand.
This article was developed in collaboration with Argenica Therapeutics, a Stockhead advertiser at the time of publishing.
This article does not constitute financial product advice. You should consider obtaining independent advice before making any financial decisions.
Related Topics

UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








