Adherium shares puff up 50pc on US approval for smart asthma inhaler

Pic: sinology / Moment via Getty Images
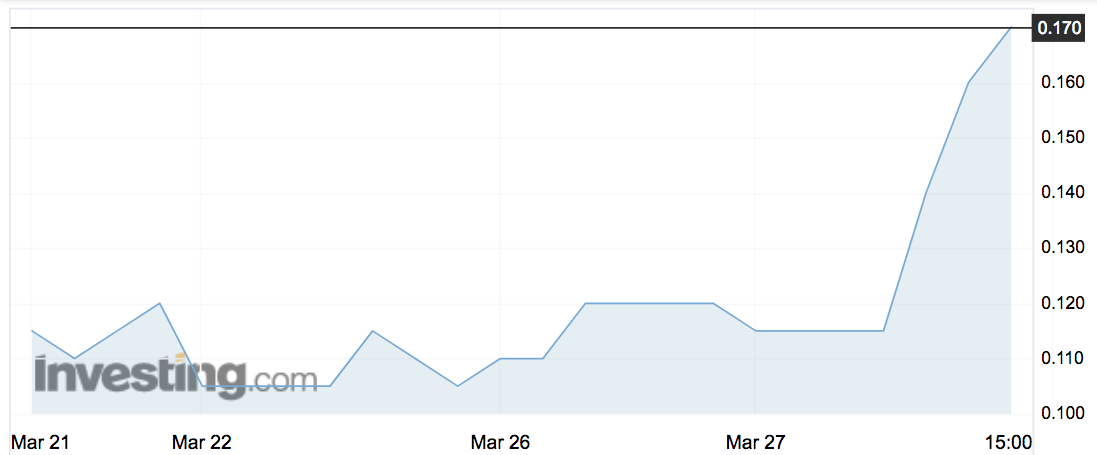
Adherium shares jumped by more than 50 per cent today after the med-tech play won US approval for retail sales of its smart asthma inhaler device.
The US Food and Drug Administration granted clearance for over-the-counter sales of Adherium’s Smartinhaler sensor.
The trade-marked Smartinhaler device is attached to a patient’s inhaler to ensure patients are taking the right amount of medication for asthma and Chronic Obstructive Pulmonary Disease.
The sensor records the date and precise time the inhaler is used and automatically transmit it to an app on a patient’s phone or tablet – storing the information for review by doctors.
“Over-the-counter clearance means that Adherium’s Smartinhaler SmartTouch for Symbicort can now be sold directly to consumers in the US, including through pharmacies and online, without the need for the patient to first obtain a prescription,” the company said.
The stock (ASX:ADR) jumped from 12c to 18.5c after the announcement — cooling slightly to 18c by Tuesday’s close.
“We are now well positioned to launch in the US, following successes in Europe and Australia,” chief Arik Anderson told the market.
“We are providing clinically proven monitoring to support patients in adopting their physicians’ guidance into their daily lives, and so close the gap between science and real-world care.”
UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








