Dr Boreham’s Crucible: Control Bionics’ world-beating wheelchair is more than a victory for mind over matter

Tim Boreham is one of Australia's best-known small-cap share analysts and business journalists.
Amid the hype about using artificial intelligence (AI) for anything from robotic call centres to writing biotechnology columns, Control Bionics’ (ASX:CBL) new chief executive Jeremy Steele is sceptical about the use of technology for technology’s sake.
That stance may seem perverse, given the company has used technology (including AI) to develop the world’s first autonomous wheelchair system called Drove, which negates the need for a joystick, mouse or keyboard.
Steele’s point is that there are many “nonsense” claims about the benefits of artificial intelligence but “solving problems with technology is not easily done”.
In the case of the Drove, there are more advanced ways to drive a wheelchair, but the company has emphasised making it as easy to use as possible.
The headline-grabbing Drove launch hopefully marks a transformational change for the company, as well as its end-users who will be free to roam from room-to-room in their houses without requiring assistance.
“When people use it, they are struck by how simple it is, but it belies the point of how difficult it is to do,” Steele says.
“We are the world’s first because it is not an easy problem to solve.”
Thought control – in a good way
At its core, Control Bionics is about enabling disabled people to use their own ‘brain- to-muscle’ electrical signals to control communication and movement.
Neuronode deploys electro-myography (EMG), which is about capturing and processing neuro-electric signals into electronic commands.
The technology also enables a person to control a cursor with their eyes and then select an item from where the cursor lands.
Control Bionics’ first devices – Neuronode and Trilogy – allow users to communicate more effectively by tapping the neural (or visual) signals sent from the brain to the muscles.
The muscles don’t have to be functioning; as long as the electrical signals are extant. Users include those with motor neuron disease, spinal cord injuries, cerebral palsy and multiple sclerosis.
The company was founded in 2005 by Peter Ford, a computer scientist and broadcaster who anchored a technology show on the US network CNN.
In his downtime he worked with a US think tank on ‘thought control’.
In 2014, Control Bionomics acquired Therapeutic Alliances Inc of the US, which owned key patents over electro-myography monitors.
In 2016, the company launched Neuronode 3, a small and non-invasive device that uses the body’s electro-myographic signals to control a mobile phone, tablet or computer.
Control Bionics listed on the ASX on December 7, 2020 at 60 cents apiece, after raising $15 million, opening at $1.12 on the day.
Changing of the guard
A former marketer, Rob Wong joined as CEO in January 2017 with a commercialisation agenda.
In September last year Wong resigned on account of a degenerative medical condition, replaced by Steele who joined on January 19 this year
Currently based in Singapore, Melburnian Steele has more than 25 years’ experience across sectors including healthcare, software, financial services and retail.
Steele was an investment banker and mergers and acquisitions adviser, but spent the last 10 years heading the Australian cardiac testing outfit Cardioscan.
“I took that company from one [geographic] market to 10, so I would like to think I have some pretty good healthcare chops these days.”
About Neuronode
Control Bionics’ core tech Neuronode uses algorithms to convert the body’s neuro-electric signals into code, which controls the devices. Other rival assistive technology devices require a keyboard, mouse, joystick, touch screen or eye-tracking to function.
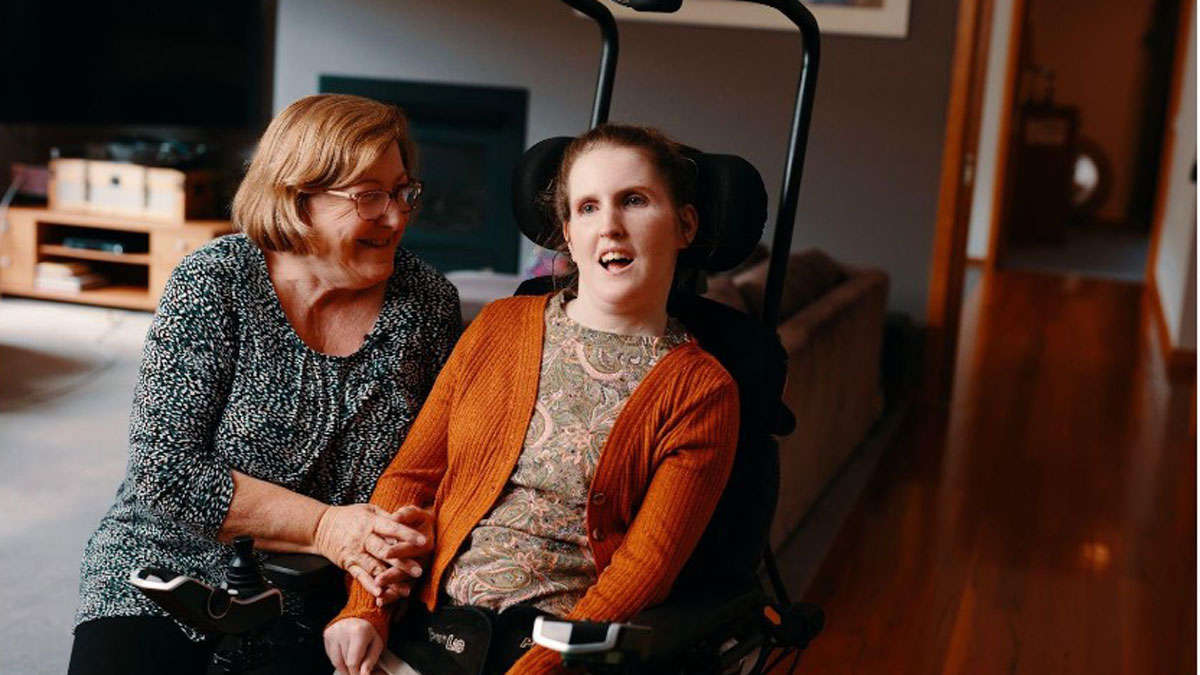
A watch-sized wireless neuro-electric sensor, Neuronode3 combines Neuronode with speech-generating software, while Neuronode Trilogy combines Neuronode with eye-tracking technology.
A key selling point is that Neuronode enables much quicker communication, which is even more important in the social media age. For instance, a user can respond instantly to a text or Snapchat message so they feel more included in the conversation.
The devices are approved in the US, European, Canada and here. The company is also tackling the Japanese market, where the products don’t need to be approved.
Drove it myself
Drove is a module that can be retrofitted to pretty much any powered wheelchair.
Developed with Deakin University’s applied artificial intelligence unit, the device enables users to move their wheelchair to specific locations within their home.
The module combines a digital camera with Control Bionics’ tracking software and the Neuronode technology.
The user gazes at a panel of rooms and selects, say, the bedroom – and away they go. An accurate path is determined by ceiling sensors, which are tracked by the Drove camera.
“If you are unable to control your wheelchair, you are stuck where you are until someone moves you,” Steele says.
It should be stressed that they cannot roam more broadly than these parameters: like a tram or train they need to stay on track. But in future it is likely they will be able to add their own ‘routes’ around the house.
Steele said the technology made a huge difference to those who tested it during development, allowing them to do simple things able-bodied people take for granted.
“It’s a level of independence they’d never considered possible,” he said. “Being able to move so the sun isn’t in your eyes – or leave a room on your terms – are things most of us don’t think twice about.”
As with autonomous vehicles, safety was a key consideration. A sensor detects obstacles and will stop the wheelchair, while the user can switch to manual mode at any time.
The size of the prize
The company won’t cite the cost of the Droves, mainly because it is still coming to the grips with the market.
As a guide, “alternate solutions for driving powered wheelchairs” retail for around $13,000 in Australia.
The company cites a total addressable market of $2 billion for the “global disability medtech space”.
In the context of Drove, about two million powered wheelchairs are sold each year.
The company estimates five to 10 per cent of these users can’t control the wheelchair themselves – or won’t be able to do so in future as their medical condition deteriorates.
“The market is significant,” Steel says. “Unfortunately, only about two per cent get access to the technology, which is heartbreakingly small.”
Reimbursement wise, Europe and US have reasonable support schemes, while locally the National Disability Insurance Scheme (NDIS) is shaping up as an important funding source.
The NDIS allocates each client a funding package, which they and their carers apply at their own discretion to approved assistive products and services.
Control Bionics’ product suite is certain to be in the mix.
The Federal Government is determined to rein-in the ballooning cost of the NDIS, but most of the tightening is expected to apply to ‘clip the ticket’ intermediaries rather than to product eligibility.
As far as selling goes, Steele says the company will tap the smarts of wheelchair and disability product distributors such as Quantum Pride and Numotion (its US reseller).
“We recognise wheelchair manufacturers and their resellers are better equipped to work with customers on installing and managing our system,” he says.
Finances and performance
Control Bionomics’ sales are still recovering from the pandemic, which greatly restricted the company’s ability to access carers and users (many of whom are immune deficient).
Nonetheless, in the December 2022 half year revenue gained 23 per cent to $2.89 million, which compares with turnover for the full 2021-’22 year of $4.47 million.
The company lost $2.96 million, compared with a $3.13 million half-year deficit previously.
About 70 per cent of revenue derived from the US, where the company is dealing with the complex tapestry of Medicare/Medicaid and private insurance funding. Having operated a US business for more than five years, Steele is confident of navigating these rules.
Control Bionics has cash of $3.02 million, enough to fund the company into 2024. But it doesn’t take a chatbot to tell us that the company will require more funding and management is mulling where these dollars might come from.
Over the last year Control Bionomics shares have traded between 37 cents (April last year) and 13 cents (June last year).
The stock peaked at $1.24 on listing day.
Apart from poor investor sentiments towards biotech stocks generally, Steele attributes the poor performance to Wong’s incapacitation – no small setback for a small company with limited management resources – and the pandemic challenges.
Undeterred, initial angel investor Phoenix Investments retains an 8.8 per cent stake.
CBL share price charts
Dr Boreham’s diagnosis:
Steele says Control Bionics works in a tricky market, “given the very nature of the people it is servicing and navigating private and public funding.”
He says the company has “established markets and attractive unit economics” – but that investors should expect some changes.
“We need to deliver consistent messaging to the market and execute and deliver against that plan,” he says. “We are formalising some strategic moves we will talk about in due course.”
Further product innovation – such as miniaturised device variants – should be expected.
“We are growing, we just need to be growing faster,” he says.
“The green shoots are there. I wouldn’t have come into the business if I didn’t think we could be wildly successful and that’s certainly my agenda.”
Given the variables of reimbursement and the rate of adoption of Control Bionics’ offerings, it’s impossible to estimate the company’s growth trajectory and profitability.
But given the benefits conferred on a large portion of the wheelchair-bound populace the company deserves every chance to be wildly successful.
As Steele says: “Giving someone independence [of movement] is a good reason for a business to exist.”
Dr Boreham is not a qualified medical practitioner. He does not possess a doctorate of any sort. This column was not written by a robot, this column was not written by a robot, this colim wash nhjj wrjkj … malfunction! malfunction!
Related Topics

UNLOCK INSIGHTS
Discover the untold stories of emerging ASX stocks.
Daily news and expert analysis, it's free to subscribe.
By proceeding, you confirm you understand that we handle personal information in accordance with our Privacy Policy.








