Lifespot hopes to sell fingerprint-activated vaping device into Israeli cannabis trials
Health & Biotech
Health & Biotech
ASX-listed Lifespot Health is focusing on Israel as a market where it hopes to sell its “vaping” device into medicinal cannabis trials.
Vaping is the process of inhaling and exhaling the vapour produced by an electronic cigarette or similar device.
Israel is regarded as one of the world’s major medical cannabis researchers and has ambitions to become a major exporter.
Last month Israel started allowing pharmacies to sell medical cannabis in a concentrated oil form to its 33,000 registered patients.
Lifespot’s vaporiser — which is activated by patient’s fingerprint — allows cannabis to be taken in the form of vapors extracted from oils or herbs.
Lifespot hopes the device — combined with the company’s BodyTel diagnostic platform — will help patients track the exact concentration used.
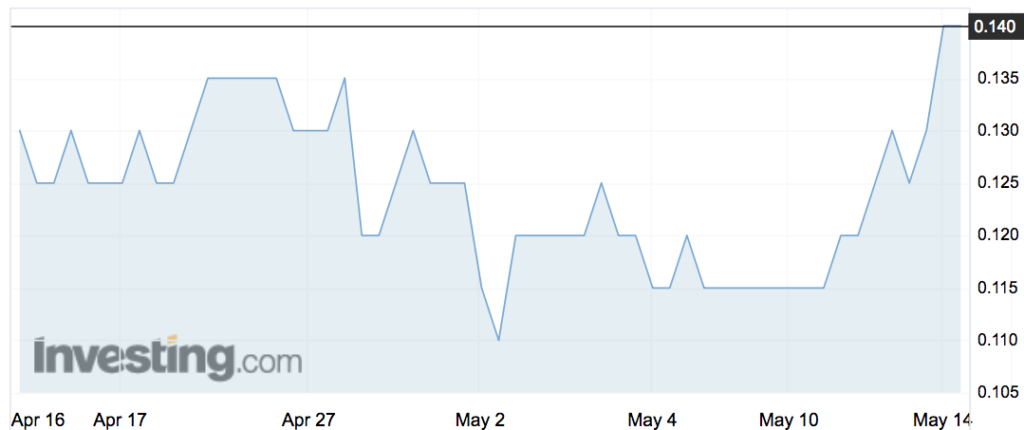
Shares in the company were up 8 per cent in early trade – at 14c.
The device has been developed by LifeSpot’s half-owned subsidiary Seng Vital.
The BodyTel platform allows patients and physicians to retrieve and evaluate readings and data collected by the vaporizser.
“We see strong potential for our integrated system as an enabler for clinical studies and commercialisation,” director Heiner Emden said.
“Patient safety, compliance and good clinical practice are very important and we are delighted to be able to provide such a high-quality system based solution to cannabis companies and healthcare professionals and be a key partner in testing and commercialisation.”
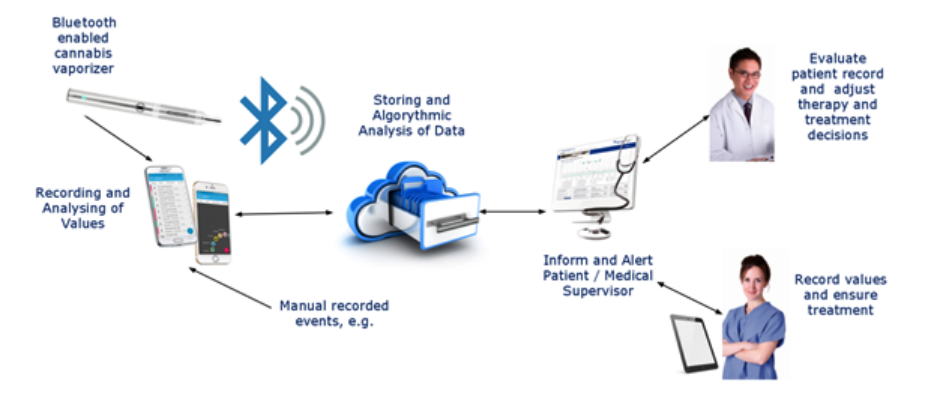 Mr Emden has previously said the device would be the “gold standard for vaporisers” with exact dosage and complete monitoring available through an integrated platform.
Mr Emden has previously said the device would be the “gold standard for vaporisers” with exact dosage and complete monitoring available through an integrated platform.
The initial “reference standard” vape is expected to be delivered to market by the end of July.
The company made $166,000 in customer receipts last quarter and had $3.9 million left in the bank.
Estimated outflows for the current quarter are $720,000.