Imugene says anti-stomach cancer treatment is safe, shares go nuts
Health & Biotech
Health & Biotech
Imugene says the first stages of a human trial for a stomach cancer treatment have shown it to be safe.
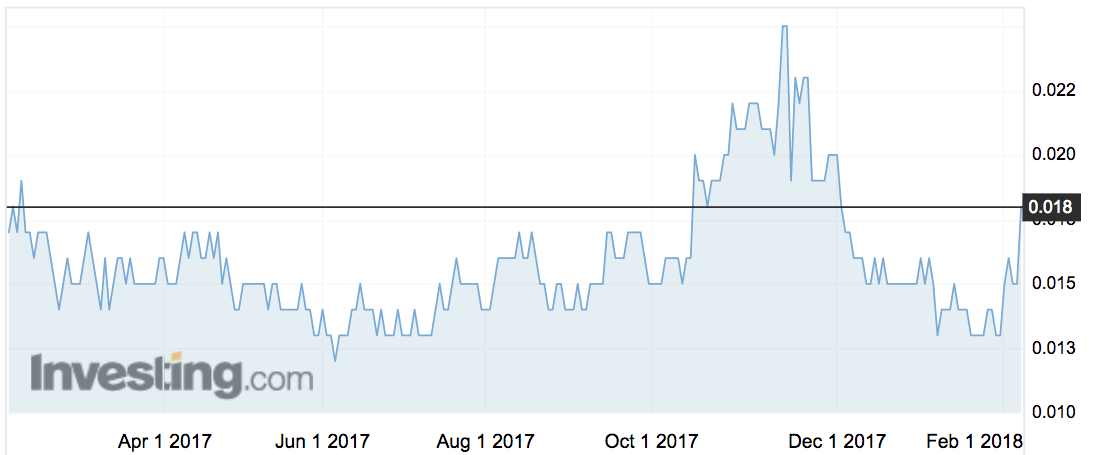
Its shares (ASX:IMU) rocketed 24 per cent on the news to 1.8c — not quite the highest it’s been over the last year (that would be 2.8c), but lifting the company’s value away from 52-week lows of 1.2c.
The next step is to up the dosage of the HER-Vaxx at the eight sites across Hong Kong, Taiwan and Thailand to see whether it decreases tumours or prolongs survival, in conjunction with chemotherapy.
“Unlike Herceptin and Perjeta, which are “injected synthetic antibody” products, HER-Vaxx activates the patient’s own immune system to produce a continuous supply of cancer targeting antibodies and induce a response against the tumors,” said lead investigator Professor Dr. Christoph Zielinski.
HER-Vaxx is a cancer immunotherapy designed to treat tumours that over-express the HER2/neu receptor, such as gastric, breast, ovarian, lung and pancreatic cancers.
The Phase 1b trial started in August last year and is testing three different doses of the HER-Vaxx vaccine with up to 18 patients.
If it’s successful, a Phase II study is intended to follow with around 68 patients with metastatic gastric cancer.
Imugene has $11.9 million in cash in order to conduct trials, after undertaking an $8.7 million capital raising late last year.
Retail shareholders weren’t convinced, however.
They only took up 23 per cent of the 1 for 21 rights issue, leaving a shortfall of just over 87 million shares.