Check Up: More clinical trials and FDA funding boost. Is this the end of biotech’s woes?
Health & Biotech
Health & Biotech
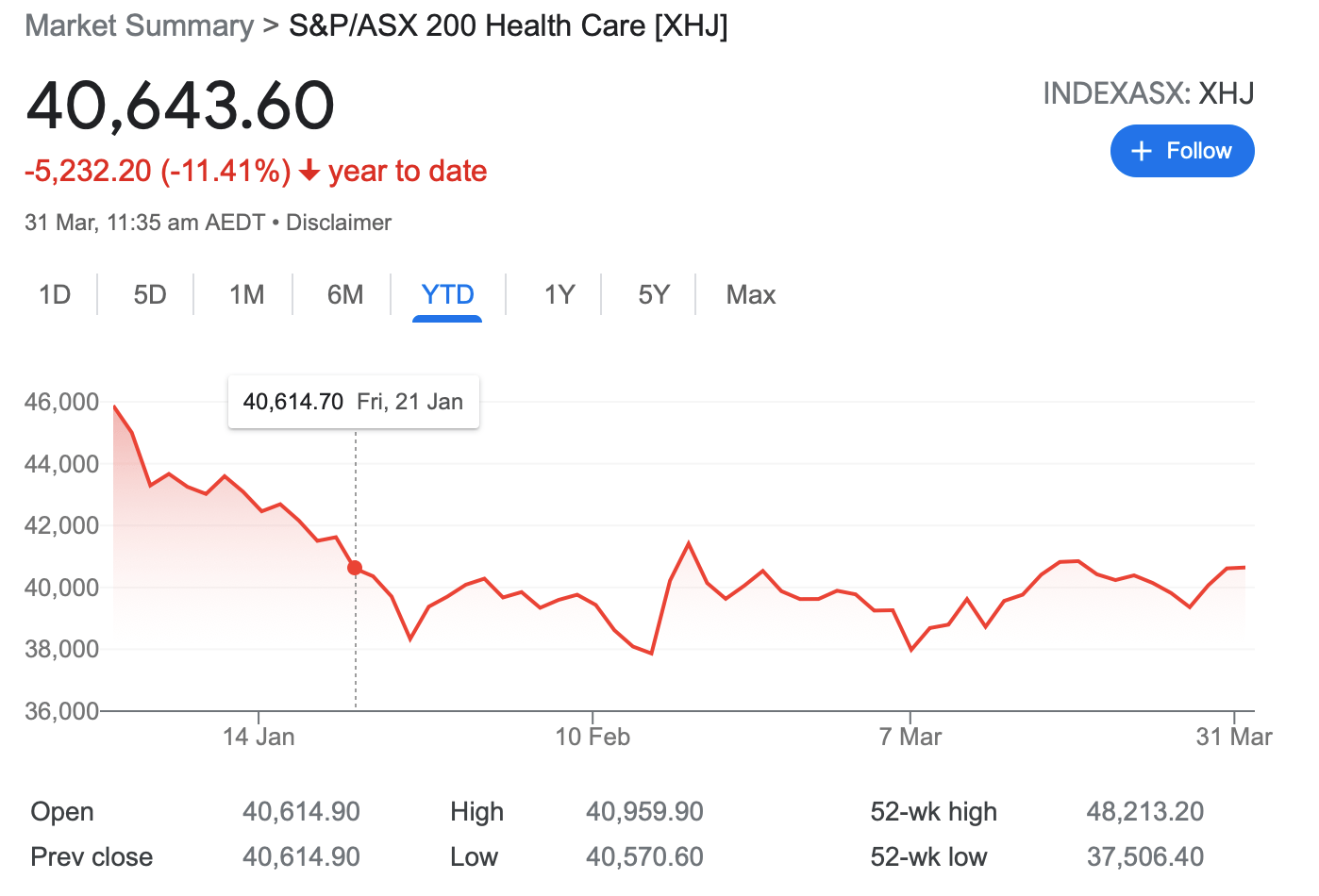
Biotech stocks entered a bear phase in the middle of 2021, and things haven’t improved much in 2022.
On the ASX, the Healthcare [XHJ] index is down 11% so far this year, which mirrors the move in the Nasdaq Biotech Index (NASDAQ:NBI).
Some analysts think it’s a part of a long term trend as we move into a higher rate cycle, but others think the sector will eventually rebound.
The reasons for the sluggish performance vary for each company, but one common denominator has been COVID-19.
A lot of development in the biotech space was slowed because of the pandemic, and this has had an impact on clinical trial registrations. There’s been a lot less clinical trial readouts over the past year, mostly caused by delays in the supply chain.
The pandemic has also made it tough for salespeople of pharma companies to meet with the doctors (their potential customers) face to face, affecting sales adversely.
Some analysts also point out there is a reduced interest in biotechnology stocks from investors in general as the pandemic wanes.
But slowly, patients are getting back to see their doctors, and hospitals are focused on clinical trials again this year.
A lot of fundies are adamant the case for investing in biotech is still a strong one, with many offering the view that biotech valuations are too low right now.
And in a sign the sector could be making a turnaround, the Biden administration has this week proposed a US$2.1 billion boost in funding for the FDA in 2023. In total, the FDA’s funding could be US$8.39 billion in 2023, a 34% increase over the US$6.25 billion for 2022.
To the ASX, here are the worst and best performing stocks this week.
| Code | Company | Price | %Week | %Month | %Year | Market cap |
|---|---|---|---|---|---|---|
| 1ST | 1St Group Ltd | 0.012 | 33% | 33% | -56% | $6,095,691.89 |
| NC6 | Nanollose Limited | 0.115 | 5% | 10% | 0% | $16,377,500.48 |
| EXL | Elixinol Wellness | 0.056 | 17% | 27% | -69% | $17,078,340.83 |
| AT1 | Atomo Diagnostics | 0.125 | 9% | -7% | -50% | $49,042,840.44 |
| MXC | Mgc Pharmaceuticals | 0.027 | 8% | 4% | -60% | $70,679,239.23 |
| PYC | PYC Therapeutics | 0.099 | 8% | 1% | -38% | $302,187,979.79 |
| CAN | Cann Group Ltd | 0.27 | 6% | 10% | -51% | $90,772,116.50 |
| PAB | Patrys Limited | 0.026 | 4% | 0% | -7% | $51,418,743.93 |
| ANP | Antisense Therapeut. | 0.13 | 0% | -10% | -35% | $83,599,247.25 |
| CPH | Creso Pharma Ltd | 0.075 | 27% | 12% | -65% | $94,614,752.65 |
| CGS | Cogstate Ltd | 2.38 | 14% | 25% | 155% | $400,403,845.38 |
| IDT | IDT Australia Ltd | 0.195 | 8% | 8% | -55% | $45,765,141.73 |
| ALC | Alcidion Group Ltd | 0.2 | 5% | 0% | -34% | $247,273,465.34 |
| IMM | Immutep Ltd | 0.39 | -4% | 13% | -6% | $329,171,129.70 |
| CDX | Cardiex Limited | 0.395 | -5% | -15% | -41% | $42,332,174.50 |
| DXB | Dimerix Ltd | 0.18 | -10% | 0% | -32% | $56,152,891.55 |
| AHK | Ark Mines Limited | 0.305 | 9% | 33% | 797% | $9,993,030.90 |
| IBX | Imagion Biosys Ltd | 0.057 | 6% | 0% | -62% | $62,788,237.90 |
| AHC | Austco Healthcare | 0.135 | 4% | 4% | 47% | $38,038,851.14 |
| AC8 | Auscann Grp Hlgs Ltd | 0.066 | 2% | -7% | -53% | $28,635,562.15 |
| 4DX | 4Dmedical Limited | 0.855 | -1% | 22% | -52% | $174,478,062.11 |
| NEU | Neuren Pharmaceut. | 4.05 | -3% | 8% | 191% | $501,343,390.48 |
| EZZ | EZZ Life Science | 0.42 | -5% | 2% | -44% | $5,231,600.00 |
| LCT | Living Cell Tech. | 0.008 | 33% | 60% | -40% | $7,198,546.06 |
| BOT | Botanix Pharma Ltd | 0.08 | 13% | 40% | -20% | $77,364,794.88 |
| PNV | Polynovo Limited | 1.145 | 4% | 15% | -59% | $747,707,489.72 |
| MDC | Medlab Clinical Ltd | 0.092 | -1% | -23% | -64% | $31,137,986.06 |
| GSS | Genetic Signatures | 1.18 | -1% | -2% | -27% | $167,459,170.32 |
| PGC | Paragon Care Limited | 0.44 | -2% | 16% | 96% | $275,082,301.98 |
| IIQ | Inoviq Ltd | 0.83 | -7% | -7% | -77% | $75,915,429.15 |
| ZNO | Zoono Group Ltd | 0.28 | 30% | 40% | -53% | $46,595,277.40 |
| EYE | Nova EYE Medical Ltd | 0.25 | 25% | -2% | -25% | $36,388,447.75 |
| KZA | Kazia Therapeutics | 1.065 | 24% | 5% | -27% | $140,619,627.59 |
| ADO | Anteotech Ltd | 0.115 | 19% | -38% | -56% | $228,507,605.90 |
| AVE | Avecho Biotech Ltd | 0.014 | 17% | 17% | -36% | $25,729,375.36 |
| NTI | Neurotech Intl | 0.059 | 16% | 31% | -26% | $41,164,248.43 |
| VHT | Volpara Health Tech | 0.84 | 15% | 15% | -36% | $211,625,468.04 |
| ZLD | Zelira Therapeutics | 0.02 | 11% | -9% | -67% | $33,502,039.28 |
| IRX | Inhalerx Limited | 0.094 | 11% | 22% | -18% | $15,835,893.96 |
| SOM | SomnoMed Limited | 1.94 | 11% | 10% | -4% | $160,553,071.10 |
| PCK | Painchek Ltd | 0.042 | 11% | 2% | -49% | $47,535,801.56 |
| MVP | Medical Developments | 3.94 | 8% | 16% | -28% | $280,843,424.58 |
| PXS | Pharmaxis Ltd | 0.09 | 7% | 2% | 13% | $49,403,084.67 |
| AN1 | Anagenics Limited | 0.045 | 7% | -8% | -44% | $9,945,958.64 |
| GLH | Global Health Ltd | 0.3 | 7% | -9% | -38% | $16,994,930.10 |
| CBL | Control Bionics | 0.385 | 7% | 7% | -35% | $19,377,398.81 |
| MEM | Memphasys Ltd | 0.07 | 6% | 17% | 4% | $55,452,483.59 |
| AMT | Allegra Orthopaedics | 0.18 | 6% | 9% | -44% | $18,802,656.54 |
| MDR | Medadvisor Limited | 0.275 | 6% | -15% | -21% | $103,983,508.48 |
| ATH | Alterity Therap Ltd | 0.02 | 5% | -5% | -38% | $48,137,491.56 |
| ARX | Aroa Biosurgery | 0.75 | 5% | -2% | -39% | $256,845,849.75 |
| VTI | Vision Tech Inc | 0.71 | 4% | 5% | -56% | $17,061,697.60 |
| CYC | Cyclopharm Limited | 1.645 | 3% | 13% | -35% | $153,134,709.72 |
| OSL | Oncosil Medical | 0.038 | 3% | 0% | -58% | $30,109,045.17 |
| ALA | Arovella Therapeutic | 0.041 | 3% | 0% | -7% | $27,463,244.27 |
| NYR | Nyrada Inc. | 0.21 | 2% | 17% | -30% | $32,761,827.00 |
| PSQ | Pacific Smiles Grp | 2.21 | 2% | -6% | -11% | $352,676,082.98 |
| OVN | Oventus Medical Ltd | 0.046 | 2% | -16% | -77% | $11,119,420.61 |
| RCE | Recce Pharmaceutical | 0.95 | 2% | -5% | -2% | $165,335,954.65 |
| RSH | Respiri Limited | 0.057 | 2% | 33% | -64% | $41,201,925.03 |
| BNO | Bionomics Limited | 0.066 | 2% | -3% | -69% | $89,321,149.10 |
| CU6 | Clarity Pharma | 0.575 | 1% | -9% | 0% | $102,671,577.95 |
| PBP | Probiotec Limited | 2.13 | 0% | -2% | -3% | $173,218,854.78 |
| BXN | Bioxyne Ltd | 0.017 | 0% | 0% | 21% | $10,882,471.77 |
| ADR | Adherium Ltd | 0.011 | 0% | -8% | -31% | $24,290,762.01 |
| ONE | Oneview Healthcare | 0.21 | 0% | -9% | -34% | $108,537,608.97 |
| TRP | Tissue Repair | 0.37 | 0% | -12% | 0% | $15,913,668.55 |
| ATX | Amplia Therapeutics | 0.145 | 0% | 0% | -43% | $28,108,830.15 |
| CAJ | Capitol Health | 0.355 | 0% | -4% | 3% | $369,987,216.76 |
| SCU | Stemcell United Ltd | 0.014 | 0% | 0% | -48% | $14,696,980.89 |
| MEB | Medibio Limited | 0.0045 | 0% | 50% | -55% | $10,786,455.55 |
| ICS | ICSGlobal Limited | 0.575313 | 0% | 0% | 7% | $6,054,604.94 |
| FFC | Farmaforce Ltd | 0.035 | 0% | 0% | -65% | $4,573,834.30 |
| CTE | Cryosite Limited | 0.47 | 0% | 4% | 84% | $22,940,494.61 |
| MX1 | Micro-X Limited | 0.21 | 0% | 27% | -40% | $96,667,051.11 |
| OIL | Optiscan Imaging | 0.145 | 0% | 7% | -38% | $89,781,912.29 |
| ACW | Actinogen Medical | 0.1 | 0% | 6% | 156% | $177,884,659.50 |
| IMC | Immuron Limited | 0.115 | 0% | 10% | -43% | $26,196,809.79 |
| GTG | Genetic Technologies | 0.005 | 0% | 25% | -44% | $46,169,825.72 |
| EPN | Epsilon Healthcare | 0.048 | 0% | -9% | -77% | $10,689,933.55 |
| S66 | Star Combo | 0.205 | 0% | -2% | -27% | $27,573,111.11 |
| DVL | Dorsavi Ltd | 0.021 | 0% | 0% | -49% | $7,431,506.99 |
| BPH | BPH Energy Ltd | 0.037 | 0% | 0% | -65% | $24,602,017.39 |
| PAL | Palla Pharma Ltd | 0.295 | 0% | 0% | -41% | $47,764,383.09 |
| IMU | Imugene Limited | 0.26 | 0% | 11% | 126% | $1,520,412,123.36 |
| 1AD | Adalta Limited | 0.076 | 0% | 3% | -55% | $23,878,040.70 |
| SHG | Singular Health | 0.205 | 0% | 5% | -65% | $13,384,259.15 |
| BIT | Biotron Limited | 0.079 | -1% | 25% | 18% | $55,452,684.33 |
| BWX | BWX Limited | 2.18 | -1% | -11% | -55% | $351,834,346.36 |
| OSX | Osteopore Limited | 0.175 | -1% | -13% | -63% | $20,521,941.65 |
| OSP | Osprey Med Inc | 0.32 | -2% | -10% | -81% | $8,210,733.44 |
| NSB | Neuroscientific | 0.285 | -2% | -3% | -3% | $40,889,418.26 |
| PAA | Pharmaust Limited | 0.094 | -2% | -6% | -6% | $29,789,764.00 |
| ACR | Acrux Limited | 0.088 | -2% | -12% | -43% | $25,030,016.09 |
| SDI | SDI Limited | 0.88 | -2% | 5% | 6% | $104,601,666.40 |
| RAP | Resapp Health Ltd | 0.082 | -2% | 22% | 19% | $70,454,160.31 |
| RNO | Rhinomed Ltd | 0.18 | -3% | -18% | 50% | $46,765,643.76 |
| RAC | Race Oncology Ltd | 2.73 | -3% | -1% | -29% | $437,084,202.68 |
| MVF | Monash IVF Group Ltd | 1.18 | -3% | 13% | 57% | $461,717,285.40 |
| VLS | Vita Life Sciences.. | 2.25 | -3% | -5% | 131% | $118,468,377.00 |
| PTX | Prescient Ltd | 0.155 | -3% | 0% | 41% | $100,764,446.78 |
| CMP | Compumedics Limited | 0.3 | -3% | -12% | -34% | $53,148,884.40 |
| HGV | Hygrovest Limited | 0.06 | -3% | 13% | -48% | $13,797,239.10 |
| CHM | Chimeric Therapeutic | 0.14 | -3% | -3% | -49% | $40,474,256.62 |
| CYP | Cynata Therapeutics | 0.42 | -3% | 2% | -31% | $60,176,169.48 |
| LBT | LBT Innovations | 0.082 | -4% | -2% | -22% | $26,231,926.61 |
| RHT | Resonance Health | 0.13 | -4% | -7% | -37% | $59,910,757.53 |
| UBI | Universal Biosensors | 0.83 | -4% | -9% | 48% | $147,730,458.32 |
| PAR | Paradigm Bio. | 1.1 | -4% | -2% | -56% | $250,355,374.50 |
| OCC | Orthocell Limited | 0.42 | -5% | 2% | -18% | $82,793,723.46 |
| RHY | Rhythm Biosciences | 1.4 | -5% | 24% | 20% | $299,710,737.20 |
| ICR | Intelicare Holdings | 0.08 | -5% | 10% | -73% | $6,910,421.12 |
| RGS | Regeneus Ltd | 0.058 | -5% | -12% | -52% | $17,773,341.01 |
| RAD | Radiopharm | 0.255 | -6% | -23% | 0% | $30,241,256.06 |
| RAD | Radiopharm | 0.255 | -6% | -23% | 0% | $30,241,256.06 |
| HCT | Holista CollTech Ltd | 0.033 | -6% | 0% | -49% | $9,086,519.87 |
| IVX | Invion Ltd | 0.016 | -6% | 0% | 78% | $102,664,218.30 |
| VBS | Vectus Biosystems | 1.3 | -6% | -5% | 7% | $45,951,530.30 |
| TD1 | Tali Digital Limited | 0.011 | -7% | -41% | -77% | $11,788,608.23 |
| TRU | Truscreen | 0.06 | -8% | -5% | -35% | $21,771,975.18 |
| BDX | Bcaldiagnostics | 0.11 | -8% | -8% | 0% | $14,472,926.82 |
| HXL | Hexima | 0.32 | -9% | -16% | 68% | $51,116,400.32 |
| ILA | Island Pharma | 0.21 | -9% | -16% | 0% | $9,081,728.88 |
| TLX | Telix Pharmaceutical | 4.56 | -9% | -8% | 4% | $1,423,839,024.00 |
| IPD | Impedimed Limited | 0.14 | -10% | -13% | 12% | $248,779,385.96 |
| NOX | Noxopharm Limited | 0.345 | -12% | 8% | -44% | $100,822,092.75 |
| JTL | Jayex Technology Ltd | 0.013 | -13% | -19% | -76% | $3,239,971.01 |
| TSN | The Sust Nutri Grp | 0.12 | -14% | -27% | -72% | $14,472,766.20 |
| ONT | 1300 Smiles Limited | 0 | -100% | -100% | -100% | $168,827,114.70 |
| AGH | Althea Group | 0.18 | -10% | 6% | -65% | $59,950,300.05 |
| IHL | Incannex Healthcare | 0.535 | -24% | -13% | 161% | $677,811,681.68 |
| ALT | Analytica Limited | 0.001 | -50% | -50% | -67% | $6,920,701.69 |
| OPT | Opthea Limited | 1.08 | 11% | 11% | -27% | $390,652,600.80 |
| PIQ | Proteomics Int Lab | 1.12 | -2% | -11% | -5% | $121,033,226.88 |
| LDX | Lumos Diagnostics | 0.395 | -6% | -21% | 0% | $61,064,676.09 |
| M7T | Mach7 Tech Limited | 0.735 | -9% | -2% | -45% | $178,767,036.00 |
| NXS | Next Science Limited | 0.89 | 1% | -1% | -30% | $189,311,120.10 |
| DOC | Doctor Care Anywhere | 0.265 | -9% | -13% | -72% | $60,781,379.43 |
Living Cell Technologies (ASX: LCT) surged 14% on Wednesday after revealing it has advanced plans for a Phase 3 clinical trial of NTCELL in Parkinson’s disease.
The company has just signed a Research Agreement with the University of Technology Sydney (UTS), and the Australian Foundation for Diabetes Research (AFDR).
The UTS agreement will allow university facilities to be used to optimise the production of NTCELL in Australia for the first time.
The NTCELL clinical trial for people with early to mid-stage Parkinson’s disease is likely to be the first xenotransplantation trial carried out in Australia, after earlier trials in New Zealand.
Cannabis company Avecho (ASX:AVE) said CBD topical therapy could become a cornerstone therapy to treat hand osteoarthritis.
On Tuesday this week, the company announced positive results from the Phase IIa study of its CBD topical gel to treat pain associated with arthritis of the hand.
During four weeks of dosing, the study demonstrated significant daily improvements in hand pain, hand functionality, grip strength, finger stiffness and anxiety.
Current treatment options for hand osteoarthritis are limited to symptom relief and preservation of hand function.
Oral administration of CBD in animal models have shown promise, but topical therapeutics like AVE’s CBD gel are of particular interest in this disease due to their targeted application and avoidance of first-pass metabolism.
Smartphone-based pain assessment company, PainChek (ASX:PCK), announced that Summerset Holdings is rolling out PainChek’s solution across all its 24 NZ care centres after a successful six-month pilot.
The contract is PainChek’s first rollout in New Zealand.
Meanwhile, AnteoTech’s (ASX:ADO) CEO Derek Thomson gave notice of his resignation as CEO of the company this week, and will serve out his six months notice period. The board has now commenced the search for a suitably experienced CEO.